Osmosis is the spontaneous net movement of solvent molecules through a selectively permeable membrane into a region of higher solute concentration. In the human body, maintaining the balance of water and solutes across cell membranes is a fundamental requirement for life. Red blood cells (RBCs), or erythrocytes, serve as a primary model for understanding these dynamics. Because the erythrocyte membrane is highly permeable to water but impermeable to many solutes, the cell’s shape and integrity are directly dictated by the osmotic pressure of the surrounding plasma. This educational illustration visualizes the physical responses of RBCs when placed in environments of varying tonicity, highlighting the concepts of crenation, equilibrium, and hemolysis.
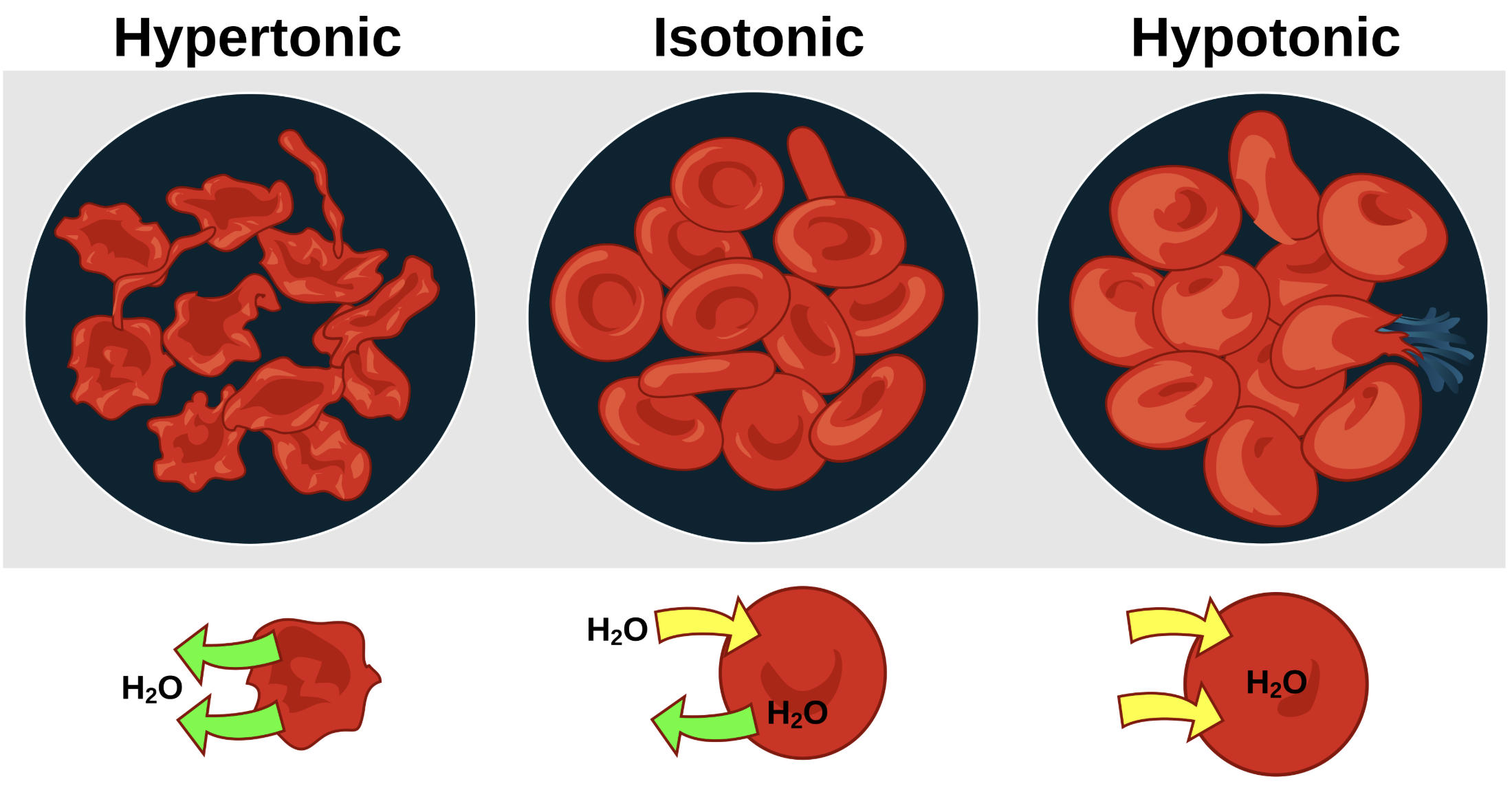
Label-by-Label Explanation
Hypertonic: This label describes a solution with a higher concentration of non-penetrating solutes compared to the interior of the cell. Under these conditions, water molecules move out of the cell via osmosis to equalize the concentration gradient, leading to a shriveled, spiked appearance known as crenation. In a clinical context, exposing cells to hypertonic solutions can impair their ability to deform and flow through narrow capillaries.
Isotonic: This label represents a state of osmotic balance where the solute concentration outside the cell equals the concentration inside. As seen in the diagram, water moves in and out of the cell at an equal rate, allowing the erythrocyte to maintain its characteristic biconcave disc shape. This morphology is essential for maximizing the surface area available for gas exchange while remaining flexible enough to traverse the microvasculature.
Hypotonic: This label identifies a solution with a lower concentration of solutes than the cell’s cytoplasm. Water enters the cell rapidly, causing it to swell and lose its central indentation. If the inflow of water exceeds the capacity of the cell membrane to expand, the cell ruptures in a process called hemolysis, which releases hemoglobin into the surrounding fluid and can lead to systemic complications.
H2O: This label represents the water molecules (solvent) moving across the semipermeable membrane. The arrows in the bottom row of the diagram illustrate the direction of net water flux in each environment: net outward flow in hypertonic, balanced flow in isotonic, and net inward flow in hypotonic solutions.
Functional Significance of Cell Morphology
The biconcave shape of the red blood cell is a masterpiece of biological engineering. This specific geometry provides a significantly larger surface-area-to-volume ratio than a sphere of the same volume, facilitating the rapid diffusion of oxygen and carbon dioxide. Furthermore, this shape provides the cell with redundant membrane area, acting as a “safety margin” that allows the cell to undergo substantial deformation and moderate swelling without immediately rupturing.
When the osmotic pressure environment shifts, this morphology is compromised. In hypertonic states, the loss of water increases the internal viscosity of the cell and makes the membrane rigid. Conversely, in hypotonic states, as the cell becomes spherical, it loses its ability to deform. Since the diameter of a red blood cell (approx. 7.5 µm) is often larger than the diameter of the smallest capillaries (approx. 5 µm), any loss of deformability significantly hinders microcirculatory perfusion.
Clinical Relevance and IV Fluid Therapy
The concepts of tonicity and osmosis are the foundation of intravenous (IV) fluid therapy. Clinicians must choose fluids based on the patient’s specific physiological needs to avoid unintended cellular damage. Standard “Normal Saline” (0.9% NaCl) is designed to be isotonic to human blood, meaning it can be used to expand the extracellular fluid volume without causing significant shifts in intracellular water or altering RBC morphology.
In cases of severe hyponatremia or cerebral edema, hypertonic solutions (such as 3% Saline) may be used to shift fluid out of cells and into the vascular compartment. Conversely, hypotonic solutions (such as 0.45% Saline or D5W) are used to provide free water to patients who are dehydrated at a cellular level. However, rapid administration of hypotonic fluids carries a high risk of causing cerebral edema and intravascular hemolysis, making precise calculation and monitoring mandatory.
Diagnostic Importance: The Osmotic Fragility Test
The sensitivity of red blood cells to osmotic changes is utilized in the diagnostic laboratory. The Osmotic Fragility Test measures the resistance of a patient’s erythrocytes to hemolysis when exposed to increasingly dilute (hypotonic) saline solutions. This is the gold-standard screening test for hereditary spherocytosis, a condition where a genetic defect in membrane proteins results in cells that are already spherical.
Because spherocytes have no redundant membrane and are already at their maximal volume-to-surface-area ratio, they rupture much more easily in slightly hypotonic environments compared to normal biconcave cells. Observing the point at which crenation gives way to rupture allows hematologists to identify underlying cytoskeletal abnormalities that might not be obvious on a standard peripheral blood smear.
Key Learning Points
- Osmosis is the movement of water from an area of low solute concentration to high solute concentration.
- Hypertonic environments cause water to leave the cell, resulting in shrunken, crenated erythrocytes.
- Isotonic environments represent dynamic equilibrium where cell volume remains constant.
- Hypotonic environments cause water to enter the cell, leading to swelling and potential hemolysis.
- Fluid therapy in clinical practice is a direct application of these osmotic principles to maintain organ perfusion and cellular integrity.
Summary
Understanding osmotic pressure is vital for any healthcare professional managing patient fluids and electrolytes. This diagram serves as a clear visual guide to the three primary states of tonicity. By recognizing how hypertonic solutions cause shriveling and hypotonic solutions cause swelling, medical learners can better appreciate the narrow physiological range the body must maintain. Proper application of these concepts ensures safe fluid resuscitation and accurate diagnosis of hemolytic disorders.
Medical Learning Tips
- Always remember that water follows the solutes; a higher external salt concentration will pull water out of the cell.
- Biconcave cells have extra membrane area to allow for some swelling, whereas spherical cells rupture almost immediately in hypotonic fluid.
- 0.9% Normal Saline is the standard isotonic IV fluid used to match human plasma osmolarity of approximately 290 mOsm/L.

