In the complex theater of cellular biology, survival is defined by the ability to move molecules exactly where they are needed, regardless of physical resistance. While passive transport methods like simple and facilitated diffusion allow substances to flow down their concentration gradients like a river moving downstream, cells frequently encounter situations where they must move materials ‘uphill.’ This effort is known as active transport. It is a vital physiological process that enables cells to maintain distinct internal environments, stockpile nutrients, and expel waste products that are already present in high concentrations outside. Without active transport, the electrical signaling in our brains and the contraction of our muscles would cease instantly. It is the energetic engine that drives the most fundamental functions of human life, transforming raw energy into biological order.
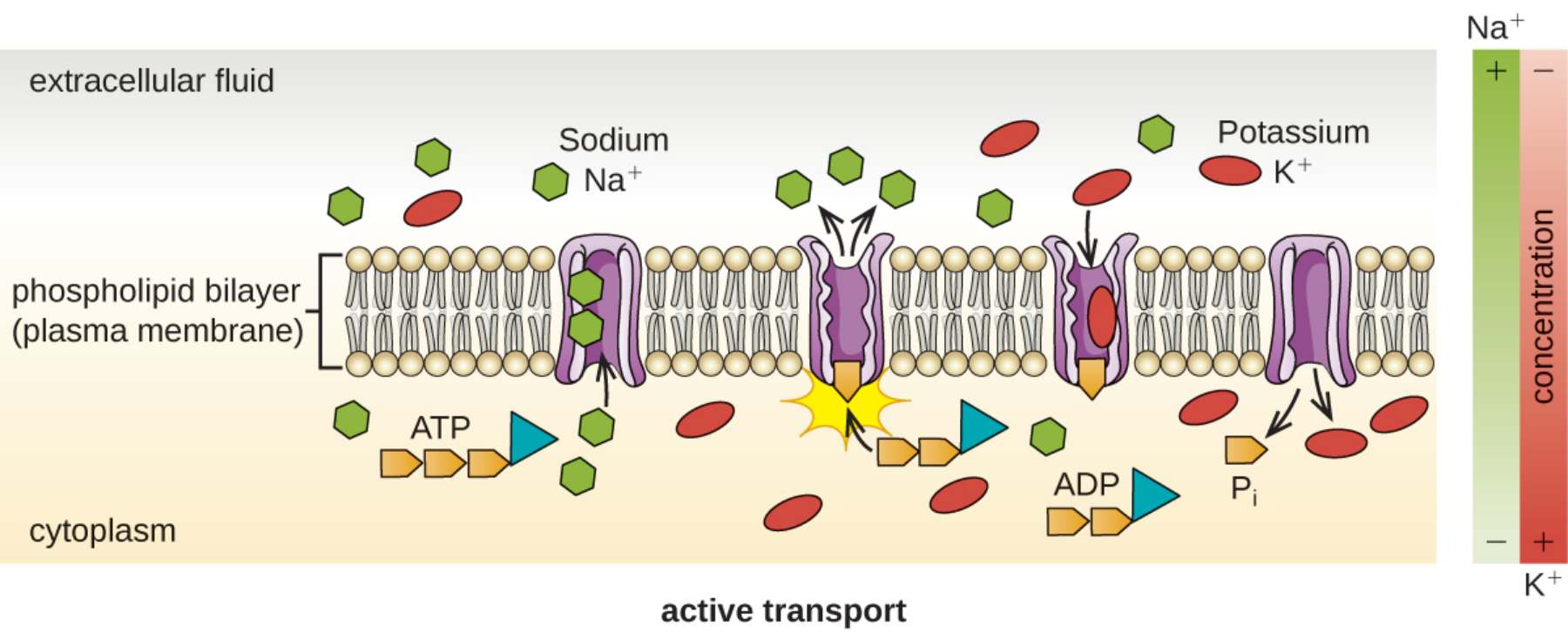
extracellular fluid: This refers to the aqueous environment surrounding the cell, characterized by high concentrations of sodium ions and low concentrations of potassium ions. It serves as the terminal location for sodium ions that are actively pumped out of the cell’s interior.
cytoplasm: The internal gel-like substance of the cell where metabolic activity is highest and specific ion balances are maintained. It is the environment from which sodium is removed and where potassium is concentrated to support cellular functions.
phospholipid bilayer (plasma membrane): The semi-permeable structural barrier that defines the cell’s boundary and restricts the free diffusion of ions. It contains specialized integral proteins, such as the sodium-potassium pump, which facilitate the transport of solutes against their gradients.
Sodium Na+: A critical cation that is kept at high levels outside the cell and low levels inside. In active transport, it is bound by carrier proteins and expelled into the extracellular fluid to maintain an electrochemical gradient.
Potassium K+: An essential ion that the cell actively pulls into the cytoplasm from the external environment. Maintaining a high internal concentration of potassium is crucial for protein synthesis and electrical stability.
ATP: Adenosine triphosphate acts as the primary energy currency that powers active transport. When it binds to a transport protein, it undergoes a chemical reaction that releases the energy required to change the protein’s shape.
ADP and Pi: These are the byproduct molecules (adenosine diphosphate and inorganic phosphate) formed after the cell harvests energy from ATP. The release of the phosphate group from the pump protein allows it to reset its configuration for the next cycle.
active transport: This overarching label describes the movement of ions or molecules across a cellular membrane from a region of lower concentration to a region of higher concentration. This process is ‘active’ specifically because it requires the direct or indirect input of metabolic energy.
concentration: The side bar represents the gradient of density for specific solutes across the membrane. It visualizes the ‘uphill’ challenge that active transport must overcome, showing higher levels of Na+ outside and higher levels of K+ inside.
The Fundamental Mechanics of Primary Active Transport
The primary form of active transport involves the direct use of chemical energy to move molecules. This energy is almost always derived from ATP hydrolysis, where a phosphate bond is broken to release kinetic power. This process is mediated by carrier proteins that function as molecular pumps. Unlike the open tunnels of channel proteins, these pumps have specific binding sites that only accept their target molecules when the protein is in a particular shape. This high degree of selectivity ensures that the cell does not waste energy moving the wrong substances or allow the internal environment to become compromised.
When the target molecules bind to the carrier protein, the energy from ATP causes the protein to undergo a conformational change. This movement physically pushes the molecules through the membrane and releases them on the other side. Because this movement is directed against the natural flow of diffusion, the cell must continuously supply energy to keep the pumps running. This is a significant metabolic investment; in many cells, up to 40% of the total energy budget is dedicated purely to maintaining these ionic gradients, highlighting how central this process is to biological survival.
The Sodium-Potassium Pump: A Biological Mainstay
The most famous and critical example of primary active transport in the human body is the sodium-potassium pump (Na+/K+-ATPase). This pump is found in the plasma membrane of virtually every cell and is responsible for maintaining the high extracellular sodium and high intracellular potassium levels seen in the diagram. For every cycle, the pump moves three sodium ions out of the cell and brings two potassium ions in. This stoichiometry is not random; because three positive charges leave for every two that enter, the pump helps create a net negative charge inside the cell.
This cycle is a beautifully orchestrated sequence of events. First, three sodium ions from the cytoplasm bind to the pump. Then, an ATP molecule is broken down, and the resulting phosphate group attaches to the pump (phosphorylation), causing it to flip its opening toward the outside of the cell. After releasing the sodium and binding two potassium ions from the exterior, the phosphate group falls off (dephosphorylation), causing the pump to snap back to its original orientation, releasing the potassium into the cell. This constant ‘pumping’ action is what allows our hearts to beat and our neurons to transmit information at lightning speed.
Secondary Active Transport and Cotransport Systems
While primary active transport uses ATP directly, secondary active transport is more subtle. It uses the energy stored in an existing concentration gradient—usually created by the primary sodium-potassium pump—to move a second substance against its own gradient. Think of it as a form of biological ‘carpooling.’ For example, many cells use the high pressure of sodium wanting to enter the cell (due to the work of the primary pump) to pull glucose or amino acids inside along with it. This is known as symport or cotransport.
This mechanism is essential in the digestive system and the kidneys. In the small intestine, sodium-glucose transporters (SGLTs) allow the body to absorb every last bit of sugar from a meal, even when glucose levels inside the intestinal cells are already quite high. By ‘hitching a ride’ on the sodium ions that are naturally diffusing into the cell, glucose can be moved effectively without a direct ATP bond being broken for each glucose molecule. This interconnectedness of transport systems demonstrates the efficiency and elegance of cellular design.
Physiological Significance and Electrochemical Gradients
The cumulative effect of these pumps is the creation of an electrochemical gradient across the cell membrane. This gradient is essentially a form of stored potential energy, similar to water behind a dam. By keeping ions unbalanced, the cell is always ‘primed’ to perform work. In the nervous system, a sudden opening of ion channels allows these gradients to collapse momentarily, creating the electrical pulse known as an action potential. Without the active transport pumps to reset these gradients afterward, a neuron could only fire once before becoming useless.
Beyond signaling, active transport is crucial for maintaining homeostasis and osmotic balance. By controlling the concentration of solutes inside the cell, active transport indirectly controls the movement of water via osmosis. If a cell were to stop pumping out sodium, the internal concentration would rise, drawing in excessive water and eventually causing the cell to swell and burst. Thus, active transport is the ultimate regulator of cellular volume and pressure, ensuring that every tissue in the body remains structurally sound and functionally capable.
Clinical Pathophysiology: When Pumps Fail
Given the importance of active transport, it is no surprise that malfunctions in these processes lead to severe clinical conditions. Certain toxins, such as those found in poisonous mushrooms or pufferfish, work by specifically blocking ion channels or pumps, leading to rapid paralysis and death. In medical practice, we sometimes intentionally manipulate these pumps. For example, cardiac glycosides (like digitalis) are used to treat heart failure by partially inhibiting the sodium-potassium pump. This leads to a slight increase in internal sodium, which eventually increases internal calcium, resulting in a stronger, more efficient heart contraction.
Furthermore, genetic mutations in transport proteins can cause a wide range of hereditary disorders. Cystic fibrosis is perhaps the most well-known, caused by a defect in an active transport channel for chloride ions. When the pump fails to move chloride properly, the osmotic balance is destroyed, leading to the thick, dehydrating mucus that characterizes the disease. Ongoing research into gene therapy and molecular pharmacology continues to target these specific protein pumps, aiming to restore the vital ‘uphill’ movement of molecules that defines a healthy human body.

