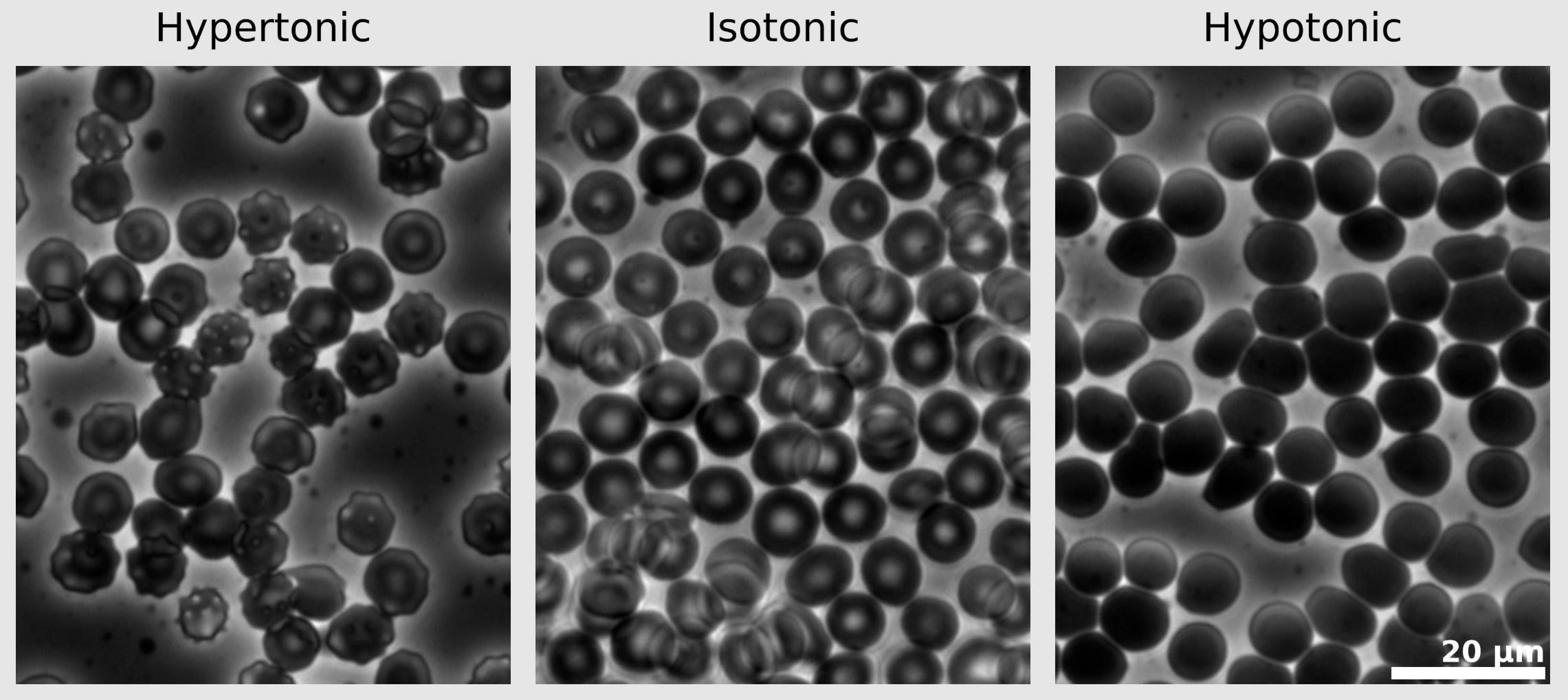
Osmosis is a fundamental physiological process that governs fluid distribution across biological membranes. In the human body, erythrocytes (red blood cells) serve as ideal biological osmometers, as their morphology is highly sensitive to the tonicity of the surrounding plasma. The provided micrographs illustrate the dramatic structural changes red blood cells undergo when exposed to hypertonic, isotonic, and hypotonic environments. Understanding these visual changes is not merely an academic exercise; it is essential for clinical practitioners involved in fluid resuscitation, electrolyte management, and the diagnosis of hematological disorders.
Label-by-Label Explanation
Hypertonic: This label identifies the micrograph where the extracellular fluid has a higher concentration of non-penetrating solutes than the intracellular fluid. Under these conditions, water moves out of the cell via osmosis, causing the cell to shrivel and develop a spiked or scalloped appearance, a process known as crenation. Clinically, this occurs when highly concentrated solutions, such as 3% saline or mannitol, are administered to draw fluid from tissues into the intravascular space.
Isotonic: This label highlights the state of dynamic equilibrium, where the solute concentration in the extracellular fluid matches that inside the red blood cell. The cells maintain their characteristic biconcave disc shape, which provides an optimal surface-area-to-volume ratio for gas exchange. Maintaining an isotonic environment (approximately 280-300 mOsm/L) is the primary goal of standard intravenous maintenance fluids like 0.9% normal saline.
Hypotonic: This label refers to an environment where the extracellular solute concentration is lower than that of the cell cytoplasm. Water rushes into the cell, causing it to swell and lose its biconcave indentation, eventually becoming spherical. If the osmotic pressure gradient is too steep, the cell membrane will rupture, leading to hemolysis and the release of intracellular contents like hemoglobin into the plasma.
20 µm: This is a scale bar representing 20 micrometers in length, providing a reference for the size of the erythrocytes. Since a typical human red blood cell is approximately 7 to 8 micrometers in diameter, this scale bar allows the observer to estimate that approximately two to three normal cells could fit across the length of the bar, confirming the magnification level of the micrograph.
Physiological Foundations of Osmosis
The movement of water across the erythrocyte membrane is regulated by the concentration of solutes that cannot cross the lipid bilayer. According to the principles of osmotic pressure, water moves from an area of low solute concentration (high water potential) to an area of high solute concentration (low water potential). The red blood cell membrane is highly permeable to water but relatively impermeable to ions like sodium and potassium, making the cell susceptible to rapid volume changes when plasma osmolarity fluctuates.
The tonicity of a solution—its ability to cause a cell to gain or lose water—depends on the concentration of non-penetrating solutes. While osmolarity refers to the total number of solute particles per liter of solution, tonicity accounts for only those solutes that exert an osmotic force. This distinction is vital in medicine; for example, urea is an osmolyte that can cross cell membranes, meaning a solution high in urea might be hyperosmotic but effectively isotonic or even hypotonic in terms of its impact on cell volume.
Morphological Responses and Clinical Implications
The three states shown in the image correlate directly to clinical scenarios. Crenation, seen in the hypertonic panel, occurs when the intravascular space becomes hyperosmolar, often due to severe dehydration or the therapeutic use of osmotic diuretics. The shrunken, spiked cells (echinocytes) have reduced flexibility, which can impair their ability to navigate the microvasculature, potentially leading to impaired oxygen delivery.
In contrast, the swelling seen in the hypotonic panel is a precursor to hemolysis. In clinical practice, the accidental infusion of large volumes of sterile water or highly dilute solutions can lead to massive intravascular hemolysis. This is a medical emergency, as the liberated free hemoglobin is nephrotoxic and can precipitate acute kidney injury. Therefore, hypotonic fluids like 0.45% saline are administered with extreme caution and usually only to correct specific free-water deficits in patients with hypernatremia.
Diagnostic Significance in Hematology
The principles of osmotic pressure are utilized in diagnostic tests such as the Osmotic Fragility Test. This test is primarily used to diagnose hereditary spherocytosis, a condition where red blood cells are abnormally spherical and less resistant to osmotic stress. In this laboratory procedure, a patient’s blood is exposed to increasingly hypotonic solutions; cells from a patient with spherocytosis will lyse at much higher salt concentrations than healthy, biconcave cells would. This highlights the importance of the biconcave shape in providing a “buffer” zone that allows for some degree of swelling before the membrane reaches its elastic limit.
Educational Summary for Medical Learners
For medical students and healthcare professionals, this image serves as a visual anchor for fluid and electrolyte physiology. It demonstrates that the physical shape of a cell is a direct reflection of its chemical environment. When reviewing a patient’s metabolic panel, one should visualize these micrographs to understand how shifts in sodium, glucose, or therapeutic infusions are affecting the cellular population. Mastery of tonicity is the bridge between basic biochemistry and the safe administration of intravenous therapy.
- Isotonic fluids (e.g., 0.9% NaCl, Lactated Ringer’s) expand extracellular volume without altering cell size.
- Hypertonic fluids (e.g., 3% NaCl, Mannitol) draw water out of cells to reduce edema or expand plasma volume.
- Hypotonic fluids (e.g., 0.45% NaCl, D5W) provide free water to the intracellular compartment but carry a risk of swelling if given too rapidly.
Medical Learning Tips
- Water always moves toward the compartment with the higher concentration of non-penetrating solutes.
- Biconcave disc shape provides a safety margin allowing RBCs to swell significantly before rupturing.
- Normal saline (0.9% NaCl) is considered isotonic because it exerts an osmotic pressure nearly identical to human plasma.

