For patients navigating the challenges of end-stage renal disease, the quality and longevity of their vascular access are fundamental to their survival and quality of life. Among the various methods used to facilitate the life-sustaining process of hemodialysis, the Arteriovenous Graft stands out as a critical surgical solution, particularly for individuals whose natural veins are not suitable for a traditional fistula. This complex surgical connection involves the use of a synthetic or biological tube to bridge the gap between a high-pressure artery and a lower-pressure vein, creating a conduit capable of handling the high blood flow rates required for modern dialysis machines. By creating this artificial shortcut, surgeons provide a durable site where dialysis needles can be repeatedly inserted without the risk of vessel collapse. Understanding the structural nuances, surgical placement, and long-term maintenance of these grafts is essential for patients, caregivers, and medical professionals alike, as it represents the literal lifeline for those whose kidneys can no longer filter toxins from the bloodstream.
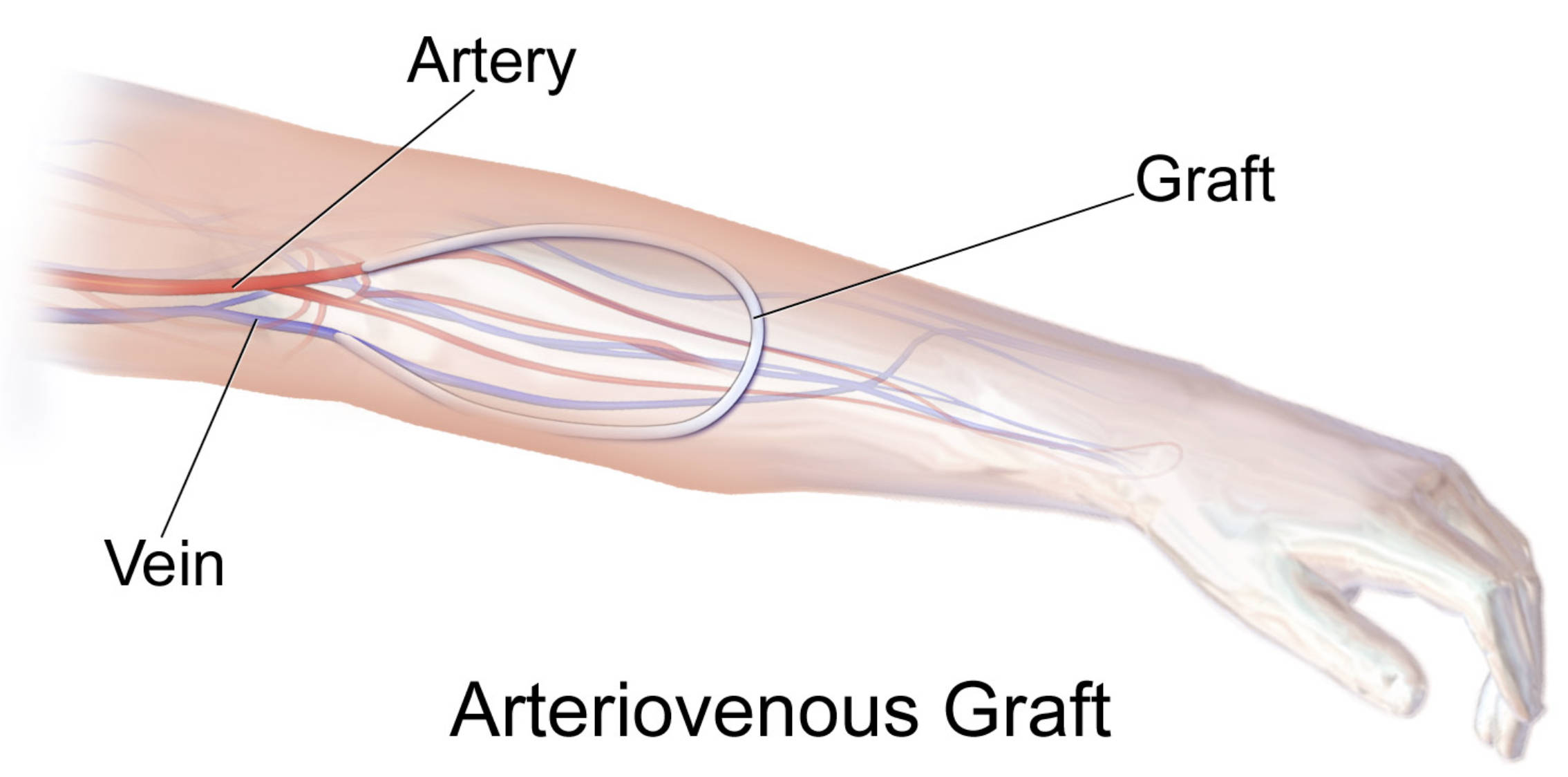
Artery: This is the high-pressure blood vessel, typically the brachial or radial artery in the arm, that serves as the inflow source for the graft. It provides the necessary volume and velocity of oxygenated blood required to power the dialysis process effectively.
Vein: This is the return vessel, such as the cephalic or basilic vein, where the blood is directed back into the systemic circulation after passing through the graft. Because the graft increases pressure within the vein, the vein wall eventually thickens to accommodate the higher flow over time.
Graft: This is a flexible, synthetic tube—most commonly made of expanded polytetrafluoroethylene (ePTFE)—that is surgically tunneled under the skin to connect the artery directly to the vein. It acts as a resilient puncture site for the large-bore needles used during hemodialysis treatments.
The Clinical Role of the Arteriovenous Graft
The primary purpose of an Arteriovenous Graft is to provide a reliable and accessible site for the repeated cannulation required during dialysis. When a patient reaches advanced stages of chronic kidney disease, their kidneys lose the ability to remove waste products and excess fluid from the blood. Hemodialysis solves this by pumping blood out of the body, through an artificial filter, and back into the circulation. However, normal veins are too small and thin-walled to support the rapid flow of blood (usually 300 to 500 mL/min) needed for this process. Furthermore, repeated needle sticks would cause a normal vein to scar and collapse within weeks.
While an arteriovenous fistula (a direct connection between an artery and a vein) is often the preferred first choice for vascular access, many patients have veins that are too narrow, scarred from previous IV use, or weakened by age. In these cases, the graft becomes the gold standard. It provides a larger surface area for needle placement and can typically be used much sooner after surgery than a fistula. For many patients with renal failure, the graft is the only viable option that avoids the high infection risks associated with long-term central venous catheters.
Surgical Anatomy and Placement Techniques
The placement of a graft is a meticulous surgical procedure usually performed under local anesthesia with sedation or general anesthesia. The surgeon identifies a suitable artery and vein, often in the non-dominant forearm or upper arm. The graft itself is a medical-grade tube that is tunneled just beneath the surface of the skin in either a “loop” or a “straight” configuration. In a loop graft, both the arterial and venous connections are made in the same general area, with the tube curving through the forearm. In a straight graft, the tube extends directly from a lower artery to a higher vein.
One of the most important aspects of the surgery is the “anastomosis,” which is the point where the graft is stitched to the natural blood vessel. The venous anastomosis is a particularly critical site, as the high-pressure blood exiting the graft can cause turbulence where it meets the vein. This turbulence can lead to the overgrowth of cells (neointimal hyperplasia), which is a leading cause of graft failure. Surgeons must carefully angle the connections to minimize this turbulence and ensure the longest possible lifespan for the access site.
Comparing Grafts with Other Access Methods
Choosing the right access method involves a balance of benefits and risks tailored to the individual patient’s anatomy. It is helpful to compare the graft to the other primary methods:
- Arteriovenous Fistula: Generally has the lowest risk of infection and the longest lifespan. However, it requires a “maturation” period of several months and may fail to develop in patients with small vessels.
- Arteriovenous Graft: Can be used within 2 to 3 weeks of surgery and is easier to place in patients with poor vascular quality. Its main drawbacks are a higher risk of clotting (thrombosis) and a slightly higher infection rate compared to fistulas.
- Central Venous Catheter: Can be used immediately but has the highest rate of life-threatening infections and can cause permanent damage to the large veins in the chest. It is usually reserved as a temporary bridge while a graft or fistula heals.
The Healing and Maturation Process
Unlike a fistula, which must “ripen” by allowing the natural vein to thicken and enlarge over several months, a graft is ready for use as soon as the surgical swelling subsides. This typically takes about two to three weeks. During this time, the tissue around the graft begins to incorporate the synthetic material, anchoring it firmly in place. Patients are advised to keep the arm elevated and avoid heavy lifting during this initial healing phase to prevent the graft from shifting or bleeding.
Medical staff will monitor the graft for a “thrill” and a “bruit.” The thrill is a distinct vibration that can be felt by touching the skin over the graft, caused by the high-velocity blood flow. The bruit is the sound of that flow, which can be heard with a stethoscope. The absence of either is a medical emergency, indicating that the graft may have clotted. Once the surgeon clears the graft for use, the dialysis team will use a specific rotating pattern for needle insertion to prevent the synthetic material from wearing out prematurely in one spot.
Daily Maintenance and Patient Care
A graft requires daily vigilance from the patient to ensure it remains functional. Because the graft is a foreign object in the body, it is more susceptible to infection than natural tissue. Patients must wash the access site with antibacterial soap before every dialysis treatment and keep the area clean and dry at home. Any signs of redness, warmth, or discharge must be reported to a nephrologist immediately.
In addition to hygiene, protecting the physical integrity of the graft is paramount. Patients are taught to avoid wearing tight clothing, jewelry, or watches on the access arm, as these can restrict blood flow and cause the graft to clot. Blood pressure readings and blood draws should never be performed on the arm with the graft. By feeling for the thrill every morning and evening, patients become the first line of defense in detecting potential issues before they become catastrophic.
Addressing Complications: Stenosis and Thrombosis
Despite best efforts, grafts are prone to certain mechanical issues. The most common is stenosis, a narrowing of the vessel, which usually occurs where the graft connects to the vein. When the vein narrows, blood flow slows down, significantly increasing the risk of thrombosis, or the formation of a blood clot. If a graft clots, the patient cannot receive dialysis, making this a critical situation. Interventional radiologists can often save a failing graft by performing an angioplasty, where a small balloon is used to stretch open the narrowed area, or a thrombectomy to remove the clot.
Another rare but serious complication is “steal syndrome.” This occurs when the graft “steals” too much blood from the hand, leading to coldness, pain, or numbness in the fingers. If left untreated, steal syndrome can lead to tissue damage. Surgeons may need to revise the graft to restrict the flow or, in severe cases, close the graft entirely to protect the limb. Understanding these risks helps patients recognize symptoms early, allowing for elective repairs rather than emergency surgeries.
Conclusion: A Vital Bridge to Health
The arteriovenous graft is a testament to the ingenuity of modern vascular surgery, providing a path to treatment for those who might otherwise have no options. While it requires more maintenance and carries higher risks than a natural fistula, its ability to provide a robust, high-flow access site makes it an indispensable tool in nephrology. By adhering to strict hygiene protocols, monitoring the site daily, and seeking early intervention for flow issues, patients can rely on their graft for years of successful dialysis. As medical technology advances, new graft materials and coatings are being developed to further reduce the risk of infection and clotting, promising an even better outlook for individuals living with kidney disease. Ultimately, the graft is more than just a piece of synthetic tubing; it is a vital bridge that connects a patient to the life-saving technology they need to thrive.

