Fungal infections often represent a unique challenge to the human immune system, particularly those caused by organisms that can seamlessly transition between environmental and parasitic forms. Among the most clinically significant of these is Histoplasma capsulatum, a fungus that thrives in the nitrogen-rich soil often associated with bat guano and bird droppings. While it resides quietly as a mold in the environment, it becomes a formidable pathogen once its microscopic spores are disturbed and inhaled by humans. This organism is the primary causative agent of histoplasmosis, a disease that ranges from a mild, self-limiting flu-like illness to a severe, life-threatening systemic infection. The ability of the fungus to adapt to the physiological temperature of the human body is the key to its survival and virulence. By understanding the growth situations and morphological shifts shown in the accompanying diagram, clinicians can better predict the progression of the disease and implement effective antifungal strategies. This article explores the intricate ecological and biological journey of the fungus, tracing its path from dark, humid attics to the complex cellular environments of the human respiratory and circulatory systems.
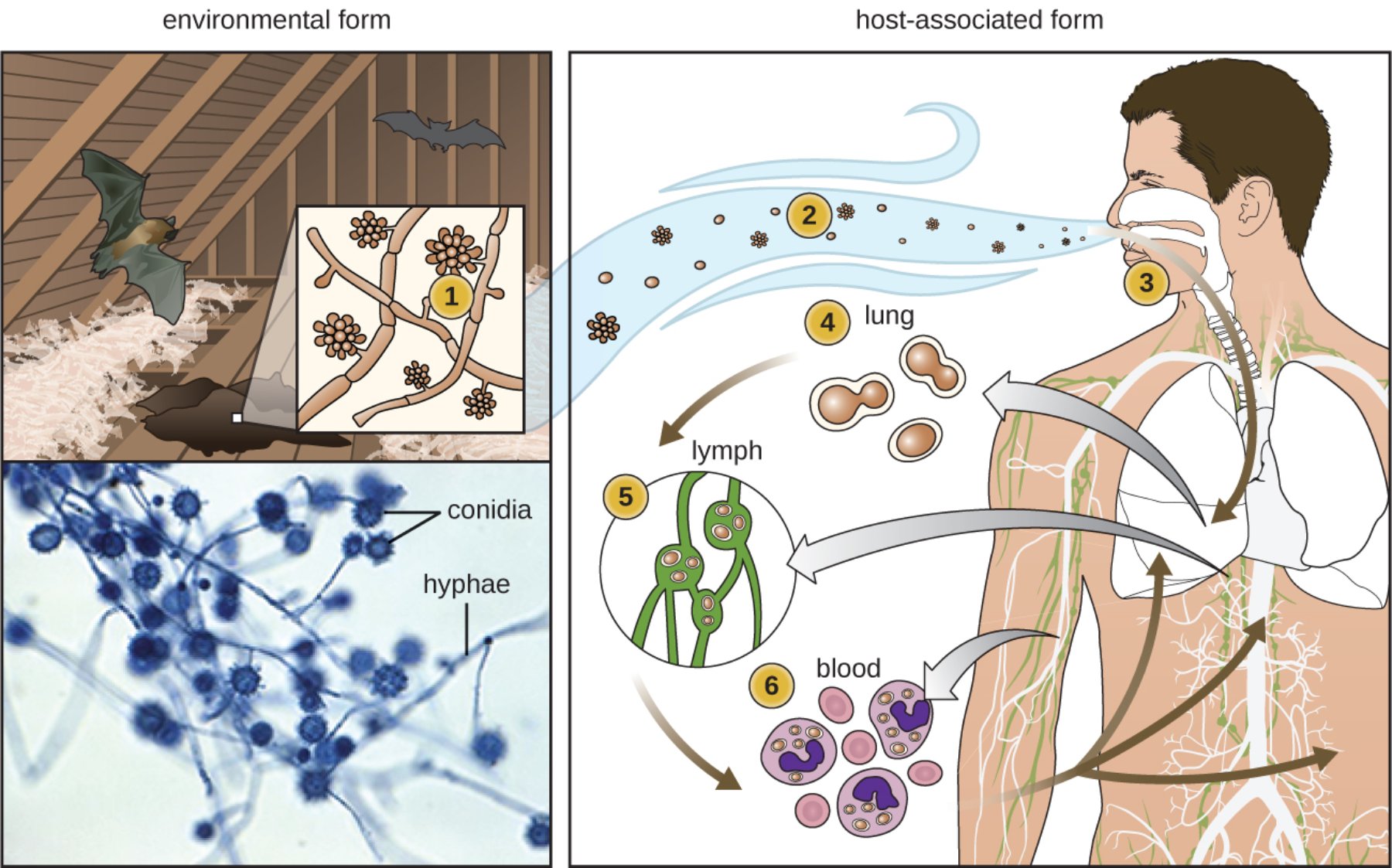
Environmental form (1): In this stage, the fungus exists as a multicellular mold characterized by branching hyphae and reproductive conidia. It typically grows in damp soil or areas contaminated with bat or bird excrement, which provide the high levels of nitrogen and phosphorus needed for its development.
Aerosolized spores (2): When the environmental source is disturbed by wind or human activity, the microconidia become airborne and can be easily inhaled. These spores are extremely small and lightweight, allowing them to remain suspended in the air for extended periods.
Inhalation (3): The microscopic conidia enter the human respiratory tract through the nose and mouth during normal breathing. Once they bypass the upper airway defenses, they travel deep into the alveoli of the lower lungs.
Lung transformation (4): Inside the warm environment of the human lung, the conidia undergo a rapid morphological change into unicellular yeast. This yeast phase is the pathogenic form of the fungus, specifically adapted to survive at the human body temperature of 37 degrees Celsius.
Lymphatic spread (5): The yeast cells are captured by immune cells and transported through the lymphatic vessels to the hilar and mediastinal lymph nodes. This step is a common precursor to the dissemination of the infection beyond the pulmonary system.
Blood dissemination (6): In some individuals, particularly those with compromised immune systems, the yeast enters the bloodstream to reach distant organs. It is typically carried within macrophages, which unfortunately serve as mobile vehicles for the fungus to reach the spleen, liver, and bone marrow.
Conidia: These are the asexual reproductive spores produced by the mold form of the fungus. They are the primary infectious particles, often identified under the microscope by their distinctive tuberculate or spiked outer walls.
Hyphae: These represent the vegetative, filamentous structures of the environmental mold. They form an extensive network known as a mycelium, which is responsible for absorbing nutrients from the soil or guano in which it grows.
The Ecological Niche and Environmental Growth
The survival strategy of the fungus is deeply rooted in its environmental surroundings. It is not found everywhere; rather, it prefers specific ecological niches characterized by high moisture and specific chemical compositions. The fungus is most commonly associated with river valleys, particularly the Ohio and Mississippi River valleys in North America. The presence of bat guano or bird droppings acts as a potent fertilizer, creating a microenvironment where the mold can flourish. In these settings, the fungus grows as a saprophyte, breaking down organic matter and producing vast quantities of microconidia and macroconidia.
Risk factors for exposure are often occupational or recreational. Farmers, construction workers, and cave explorers (spelunkers) are at the highest risk because their activities often disturb the soil or guano where the fungus resides. When an old attic is cleaned or a cave is explored, millions of spores can be kicked up into the air. Interestingly, birds themselves do not carry the disease because their high body temperature prevents the fungus from transitioning to the pathogenic yeast phase; they merely provide the nutrient-rich droppings that allow the mold to grow in the soil below.
Pathogenesis: The Switch to the Yeast Phase
The most remarkable biological feature of this organism is that it is a dimorphic fungus. Dimorphism is the ability to exist in two different structural forms depending on environmental cues, primarily temperature. In the soil at 25°C, it remains a mold. However, as soon as the conidia are inhaled and reach the 37°C environment of the human lung, a dramatic genetic reprogramming occurs. The fungus stops producing hyphae and begins to grow as a small, budding yeast. This transition is not merely a change in shape; it involves a complete overhaul of the cell wall composition and metabolic pathways to withstand the host’s internal environment.
Once the yeast form is established, it encounters the host’s primary immune defenders: the alveolar macrophages. Unlike many other pathogens that are killed upon being swallowed by these immune cells, the fungus has evolved ways to survive inside them. It can manipulate the pH of the macrophage’s internal compartments, preventing the enzymes that would normally digest it from functioning correctly. By living inside the host’s own cells, the fungus effectively hides from antibodies and other circulating immune factors, allowing it to multiply and eventually spread throughout the body.
The Clinical Spectrum of Histoplasmosis
The severity of Histoplasmosis is largely determined by two factors: the intensity of the initial spore exposure and the underlying health of the patient’s immune system. Most healthy individuals who inhale a small number of spores will remain asymptomatic or experience only mild, flu-like symptoms that resolve without treatment. In these cases, the body eventually walls off the infection, leaving behind small calcified nodules in the lungs or spleen that can often be seen on X-rays years later.
However, if a large dose of spores is inhaled, acute pulmonary disease can occur, characterized by high fever, chest pain, and a dry cough. In chronic cases, the infection can mimic tuberculosis, causing progressive lung damage and cavities. The most feared form is disseminated disease, where the fungus spreads to the liver, spleen, skin, and even the central nervous system. This form is almost exclusively seen in immunocompromised individuals, such as those with HIV/AIDS or those taking immunosuppressive medications, and it requires aggressive medical intervention to prevent a fatal outcome.
Diagnostic Procedures and Laboratory Findings
Diagnosing an infection involves a combination of imaging, laboratory tests, and clinical history. On a chest X-ray or CT scan, the infection may appear as patchy infiltrates or small nodules. However, because these findings are non-specific, definitive diagnosis requires identifying the fungus itself. The gold standard is a fungal culture of the blood, sputum, or bone marrow, but this can take several weeks because the fungus grows slowly in the lab. Consequently, clinicians often rely on more rapid methods to begin treatment quickly.
Antigen testing is one of the most useful rapid tools. The fungus sheds certain proteins into the host’s blood and urine, which can be detected via enzyme immunoassay. Additionally, microscopic examination of tissue biopsies using specialized stains, such as the Gomori Methenamine Silver (GMS) stain, can reveal the characteristic small, intracellular budding yeast. These yeast cells are typically found clustered inside white blood cells, providing a visual confirmation of the parasitic phase shown in the growth situation diagram.
Treatment Guidelines and Antifungal Selection
Management of the infection is tailored to the severity of the symptoms. For mild cases in healthy patients, treatment is often not required, as the immune system is capable of clearing the fungus on its own. However, for those with persistent symptoms or moderate disease, the antifungal of choice is usually itraconazole. This medication is taken orally for several weeks or months and works by inhibiting the synthesis of ergosterol, a vital component of the fungal cell membrane.
In severe or disseminated cases, more intensive therapy is needed. Patients are often hospitalized and started on intravenous amphotericin B. This is a potent medication that acts quickly to reduce the fungal burden but can have significant side effects, particularly on the kidneys. Once the patient is stabilized, they are typically switched to a long-term course of oral itraconazole to ensure the infection is completely eradicated. For patients with advanced HIV, lifelong suppressive therapy may be necessary to prevent the infection from returning once the initial treatment is finished.
Prevention and Public Health Measures
Preventing infection is largely a matter of environmental awareness and personal protection. In areas where the fungus is endemic, individuals should avoid activities that stir up dust in attics, caves, or old buildings where birds or bats may have nested. If such activities are unavoidable, the use of specialized respiratory protection, such as N95 masks, is essential. Simply wearing a standard cloth mask is often insufficient to filter out the microscopic conidia that are only a few micrometers in diameter.
Public health efforts also focus on site remediation. Before cleaning an area contaminated with guano, it is often recommended to soak the material with water or a bleach solution to prevent the spores from becoming aerosolized. Additionally, educating at-risk populations—particularly those with weakened immune systems—about the dangers of “cave disease” is a critical component of preventative medicine. By respecting the ecological niche of this organism and taking proper precautions during environmental disturbances, the incidence of severe pulmonary and disseminated disease can be significantly reduced.
Conclusion: A Symbiosis of Environment and Host
In summary, the growth situation of Histoplasma capsulatum illustrates the remarkable complexity of fungal pathogenesis. The journey from a saprophytic mold in a dark attic to an intracellular parasite in the human lung is a masterclass in biological adaptation. The temperature-dependent switch to the yeast phase allows the fungus to utilize the human body as a protective environment, while the use of macrophages as transport vehicles enables it to reach every corner of the host. While most people recover without complication, the potential for severe disease reminds us of the power of microscopic organisms. Through a combination of environmental vigilance, rapid diagnostic testing, and targeted antifungal therapy, we can manage the risks associated with this ancient fungus. As we continue to expand our understanding of dimorphic fungi, the lessons learned from the lifecycle of this organism will remain foundational to the fields of mycology and infectious disease, ensuring that we are better prepared for the challenges posed by the unseen world around us.
Histoplasma capsulatum, histoplasmosis, dimorphic fungus, bat guano, microconidia, mycology, pulmonary infection, systemic mycosis, antifungal, yeast transformation

