The health of the human vascular system is fundamental to the longevity and function of every organ in the body. While we often view blood vessels as simple conduits for transport, they are in fact dynamic, multi-layered organs capable of sensing pressure, responding to chemical signals, and undergoing profound structural remodeling in response to disease. One of the most significant and clinically relevant forms of this remodeling is Arterial Calcification, a process where mineral deposits accumulate within the vessel walls. Historically viewed as a passive, inevitable consequence of aging, modern science now recognizes this as an active, highly regulated biological process akin to bone formation. When viewed under a microscope using traditional staining techniques, the transition from healthy elastic tissue to rigid mineralized plaque becomes starkly apparent. This microphotography provides a window into the silent progression of cardiovascular disease, revealing how the delicate architecture of the vessel is compromised long before a clinical event such as a heart attack or stroke occurs. By studying these histological changes, we can better understand the mechanisms of vascular stiffness and the complex interplay between inflammation and mineralization.
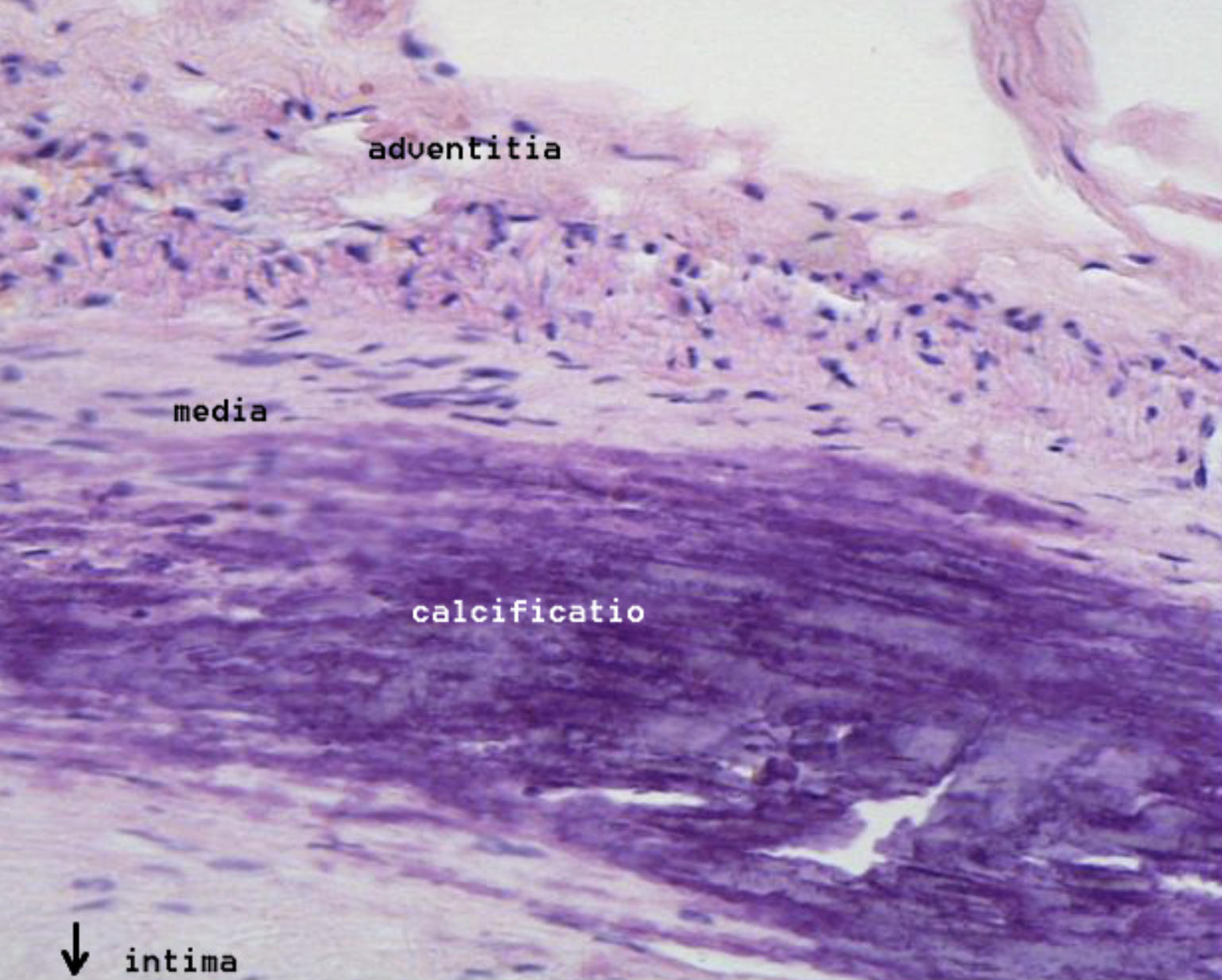
adventitia: This is the outermost layer of the arterial wall, composed of loose connective tissue, collagen fibers, and elastic filaments. It provides structural support to the vessel and contains the vasa vasorum, which are tiny blood vessels that supply oxygen and nutrients to the outer layers of the artery.
media: The tunica media is the middle layer of the vessel, primarily consisting of smooth muscle cells and varying amounts of elastic tissue. In the context of this image, the media is adjacent to the calcified region and is the primary site where the vessel\’s mechanical tone and diameter are regulated.
calcificatio: This label points to the large, darkly stained purple mass within the vessel wall, representing the accumulation of calcium phosphate minerals. This mineralization signifies an advanced stage of vascular disease, where the soft atherosclerotic plaque has transformed into a rigid, bone-like structure.
intima: The tunica intima is the innermost layer of the artery, normally consisting of a single layer of endothelium and a thin subendothelial space. In this pathological specimen, the intima is severely thickened and distorted by the presence of the atherosclerotic plaque and the underlying calcified deposits.
Histological Layers of the Arterial Wall
To appreciate the severity of vascular disease, one must first understand the normal microscopic organization of a healthy artery. The wall is essentially a sandwich of three distinct tunics. The innermost layer, the intima, is the only part of the vessel in direct contact with the circulating blood. It is lined by a specialized layer of cells that acts as a gatekeeper, controlling which molecules can pass from the blood into the deeper tissues of the wall. When this layer is healthy, it prevents blood clotting and maintains a smooth surface for laminar flow.
The middle layer, or media, is usually the thickest part of a large artery. It is dominated by smooth muscle cells that are arranged in a circular fashion. These cells are responsible for vasoconstriction and vasodilation, allowing the body to redirect blood flow to where it is most needed. The outermost layer, the adventitia, serves as the anchor. It blends into the surrounding connective tissue and contains nerves that control the smooth muscle of the media. In the microphotography provided, we see how the orderly arrangement of these layers is completely disrupted by the invasion of a mineralized plaque.
The Pathogenesis of Atherosclerotic Plaques
The journey toward mineralization typically begins with the development of atherosclerosis, a chronic inflammatory condition. It often starts with microscopic damage to the inner lining of the vessel. This damage allows low-density lipoprotein (LDL) cholesterol to seep into the subendothelial space of the intima. Once trapped, these lipids become oxidized, triggering an immune response. White blood cells, known as monocytes, are recruited to the site, where they transform into macrophages and begin consuming the oxidized lipids.
These lipid-laden immune cells are called foam cells, and they form the initial fatty streak. As the process continues, smooth muscle cells from the media migrate into the intima to wall off the fatty area, creating a fibrous cap. If the inflammation persists, the core of this plaque can become necrotic. The body\’s attempt to heal this chronic inflammation eventually leads to the recruitment of minerals, turning a soft, unstable lipid plaque into the hard, calcified mass seen in the histological slide. This represents a crossroads in the disease process, marking a shift from a predominantly lipid-driven condition to a mineral-driven structural failure.
The Mechanism of Medial and Intimal Calcification
In medical pathology, we distinguish between two main patterns of calcification. The first, intimal calcification, occurs as part of the atherosclerotic process described above. It usually forms patchy, irregular chunks of mineral that can cause the plaque to become brittle and prone to rupture. The second pattern, known as Mönckeberg\’s medial calcification, occurs within the smooth muscle layer of the media itself. This is often seen in elderly patients or those with chronic kidney disease and diabetes, leading to a profound stiffening of the vessel without necessarily blocking the flow of blood.
The actual process of mineralization is fascinating and involves a phenotypic switch in the smooth muscle cells. Under certain stressful conditions—such as high levels of phosphate in the blood or chronic oxidative stress—the muscle cells in the media begin to behave like bone-forming cells (osteoblasts). They start secreting a matrix that promotes the deposition of hydroxyapatite, the same mineral found in our bones and teeth. This is why the calcified area in the H&E stain appears deep purple; the Hematoxylin dye has a high affinity for calcium deposits, highlighting the extent of the mineralized landscape within the vessel.
Clinical Consequences of Arterial Stiffness
The transition from a flexible vessel to one containing stone-like deposits has severe mechanical consequences. Healthy arteries are compliant; they expand during the heart\’s contraction to absorb the pressure pulse and recoil during the rest phase to push blood forward. When the wall becomes calcified, this compliance is lost. The vessel becomes a rigid pipe, leading to an increase in systolic blood pressure and a decrease in diastolic pressure, a condition known as widened pulse pressure. This puts an immense strain on the heart, which must now pump against much higher resistance.
Furthermore, calcification within an atherosclerotic plaque creates “stress concentrations” at the interface between the hard mineral and the soft lipid core. During every heartbeat, the physical tug-of-war between these two materials can cause the fibrous cap of the plaque to tear. Once the cap ruptures, the highly thrombogenic interior of the plaque is exposed to the blood, leading to the rapid formation of a clot. This is the underlying mechanism for most acute myocardial infarctions and ischemic strokes. Thus, what we see under the microscope as a purple mass is not just a mineral deposit; it is a mechanical time bomb.
Diagnostic and Therapeutic Approaches
Because calcification is highly dense, it can be detected through various imaging modalities. The most common is the coronary artery calcium (CAC) score, performed via a specialized CT scan. This non-invasive test quantifies the amount of calcium in the heart vessels and is one of the strongest predictors of future cardiovascular events. In the clinical lab, researchers use microscopic analysis of biopsies, much like the image provided, to test new drugs that might inhibit the osteogenic transformation of vascular cells.
Currently, there are no approved medications that can reliably “dissolve” existing arterial calcium. Treatment instead focuses on prevention and stabilization. Statins are used to lower LDL cholesterol and stabilize the lipid core of the plaque, while blood pressure management reduces the mechanical fatigue on the vessel wall. Emerging therapies are exploring the role of Vitamin K2 and magnesium, which may help direct calcium toward the bones and away from the soft tissues of the heart. As our understanding of the cellular pathways leading to mineralization grows, we hope to move from merely observing these purple masses under a microscope to preventing their formation entirely.
Conclusion: Integrating Histology and Patient Care
The microphotography of the arterial wall serves as a powerful reminder of the physical reality of vascular disease. Behind every statistic about heart disease is a structural change occurring at the microscopic level—a disruption of the intima, a transformation of the media, and the inexorable progression of calcification. By studying the relationships between the adventitia, media, and intima, we can appreciate the exquisite design of the human body and the catastrophic consequences when that design fails. The presence of mineral deposits in a vessel is not merely a sign of age; it is a sign of a complex biological battle being lost. As medical science advances, the goal remains to keep our vessels as flexible as they were in youth, ensuring that the heart\’s vital work can continue unhindered by the development of rigid, calcified plaques. Understanding these histological details is the first step toward better diagnostic tools, more effective treatments, and a clearer path toward vascular health for all.
Arterial Calcification, Atherosclerosis, Tunica Media, Intima, Adventitia, Vascular Histology, H&E Stain, Calcified Plaque, Cardiovascular Disease, Pathophysiology

