A carotid body tumor, also recognized clinically as a chemodectoma or a glomus caroticum tumor, represents a rare but fascinating neuroendocrine neoplasm. These tumors arise from the paraganglia located at the carotid bifurcation, which are specialized clusters of chemoreceptor cells responsible for sensing changes in arterial blood gases, particularly oxygen, carbon dioxide, and pH levels. While most of these growths are benign and slow-growing, their intimate proximity to the internal and external carotid arteries, as well as several cranial nerves, presents significant diagnostic and surgical challenges. Histologically, these tumors exhibit a highly characteristic architecture that serves as a hallmark for pathologists. Understanding the microscopic nuances of a carotid body tumor is essential for differentiating it from other cervical masses and for planning precise therapeutic interventions that minimize neurovascular morbidity.
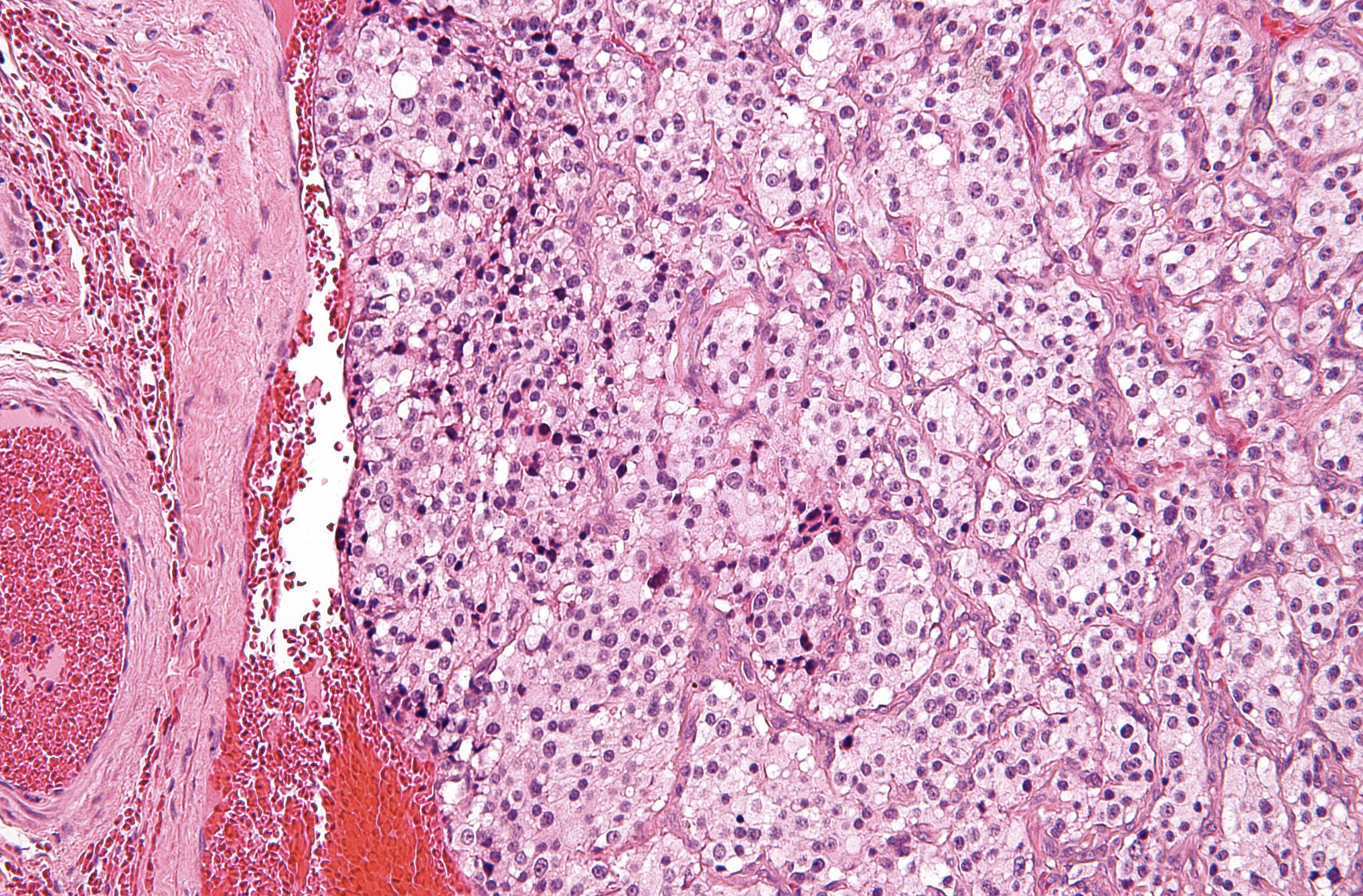
Zellballen Pattern: This term refers to the characteristic organoid or nested arrangement of cells seen throughout the tissue sample. These nests are separated by a fine, delicate fibrovascular stroma that gives the tumor its distinct mosaic appearance under the microscope.
Chief Cells: These are the predominant neoplastic cells found within the Zellballen nests, characterized by their polygonal shape and abundant granular cytoplasm. They contain neurosecretory granules that can be highlighted using specific immunohistochemical markers like synaptophysin or chromogranin.
Sustentacular Cells: These are specialized supporting cells located at the periphery of the cell nests, often appearing spindle-shaped or flattened. Although they are difficult to distinguish on standard H&E staining, they are crucial for the structural integrity of the paraganglion tissue.
Rich Vascular Stroma: The image displays an extensive network of thin-walled capillaries and blood vessels surrounding the cellular clusters. This high degree of vascularity explains why these tumors often present as pulsatile masses and carry a high risk of intraoperative bleeding.
The Anatomy and Physiology of the Carotid Body
To appreciate the pathology of a carotid body tumor, one must first understand the normal anatomy of the carotid body. Situated within the adventitia of the carotid bifurcation, the carotid body is the largest collection of paraganglia in the head and neck region. Its primary physiological role is to act as a peripheral chemoreceptor. When oxygen levels in the blood drop (hypoxia), or when carbon dioxide levels rise (hypercapnia), the carotid body triggers a reflex increase in heart rate and respiratory depth to maintain homeostasis.
- Innervation: The carotid body is primarily innervated by the Hering’s nerve, a branch of the glossopharyngeal nerve (CN IX).
- Blood Supply: It receives a rich arterial supply via the glomic artery, which usually branches directly from the external carotid artery.
- Hyperplasia: Chronic exposure to hypoxia, such as living at high altitudes or having chronic obstructive pulmonary disease (COPD), can lead to physiological hyperplasia of the carotid body cells.
Histopathological Characteristics and Differential Diagnosis
Microscopically, the carotid body tumor is a classic example of a paraganglioma. The “Zellballen” (German for “cell balls”) pattern is the most defining feature. This pattern consists of clusters of round-to-oval chief cells surrounded by a single layer of sustentacular cells. The chief cells often display “salt-and-pepper” chromatin, which is a common feature of neuroendocrine cells where the chromatin is finely clumped. While the architecture is usually consistent, some tumors may show cellular pleomorphism or nuclear atypia; however, these features do not necessarily indicate malignancy.
Pathologists often utilize immunohistochemistry (IHC) to confirm the diagnosis. Chief cells typically express neuroendocrine markers such as Synaptophysin, Chromogranin A, and CD56. Conversely, the sustentacular cells are positive for S100 protein, which helps delineate the edges of the Zellballen nests. The differential diagnosis for a mass in this region includes cervical lymphadenopathy, schwannomas, neurofibromas, and branchial cleft cysts. The presence of the nested Zellballen pattern and the specific location at the carotid fork are the primary factors in ruling out these other conditions.
Clinical Presentation and the Shamblin Classification
Most patients with a carotid body tumor present with a slow-growing, painless mass in the upper lateral neck. A unique clinical feature is Fontaine’s sign, where the mass is mobile in a lateral direction but fixed vertically because of its attachment to the carotid vessels. Large tumors may compress adjacent cranial nerves, leading to symptoms like hoarseness (vagus nerve), dysphagia (glossopharyngeal nerve), or tongue deviation (hypoglossal nerve). Approximately 5% to 10% of these tumors are functional, meaning they secrete catecholamines, leading to symptoms like hypertension, palpitations, and sweating.
To aid in surgical planning, the Shamblin classification system is used to categorize these tumors based on their relationship with the carotid arteries:
- Group I: Small tumors that are minimally attached to the carotid vessels and are easily excised.
- Group II: Moderately sized tumors that partially surround the carotid arteries but can be separated with careful dissection.
- Group III: Large tumors that intimately encase the internal and external carotid arteries, often requiring vascular reconstruction during removal.
Genetic Predisposition and Hereditary Syndromes
While the majority of carotid body tumors are sporadic, up to 35% are associated with hereditary syndromes. The most common genetic link involves mutations in the succinate dehydrogenase (SDH) gene complex, particularly subunits SDHD, SDHB, and SDHC. Patients with these mutations are more likely to develop multiple paragangliomas, often occurring bilaterally. The SDHD mutation follows a unique pattern of genomic imprinting, where the disease is usually only expressed when the mutation is inherited from the father.
In addition to SDH mutations, carotid body tumors can be associated with other hereditary conditions such as von Hippel-Lindau (VHL) syndrome, Multiple Endocrine Neoplasia type 2 (MEN2), and Neurofibromatosis type 1 (NF1). Genetic counseling and screening are strongly recommended for any patient presenting with a paraganglioma, especially if they have a family history of similar tumors or if the tumor is multifocal.
Diagnostic Imaging and Management Strategies
Imaging plays a pivotal role in the management of carotid body tumors. Duplex ultrasound is often the first-line modality, showing a hypervascular mass at the bifurcation. Computed Tomography (CT) and Magnetic Resonance Imaging (MRI) provide more detailed anatomical information. On MRI, these tumors typically show a “salt-and-pepper” appearance on T2-weighted images due to high-flow vascular voids. Digital Subtraction Angiography (DSA) remains the gold standard for assessing the blood supply and identifying the “Lyre sign,” which is the widening of the angle between the internal and external carotid arteries.
The primary treatment for carotid body tumors is surgical resection. Due to the high vascularity, preoperative embolization is often performed 24 to 48 hours before surgery to reduce intraoperative blood loss. In cases where the tumor is too large or involves critical structures, radiotherapy may be considered as a primary treatment to arrest tumor growth, although it does not usually eliminate the mass. Long-term follow-up is necessary for all patients, as late recurrences can occur, and the potential for malignancy—defined only by the presence of distant metastasis—remains a concern in roughly 5% to 10% of cases.
Conclusion and Prognostic Outlook
The prognosis for individuals with a carotid body tumor is generally excellent when the tumor is caught early and managed by an experienced multidisciplinary team. Modern surgical techniques and preoperative imaging have significantly reduced the risk of strokes and nerve injuries. However, the complexity of these tumors demands a thorough understanding of their histological nature and anatomical relationships. By combining clinical expertise with advanced pathological insights, healthcare providers can ensure optimal outcomes for patients facing this rare and intricate diagnosis.

