An arteriovenous fistula (AVF) represents a significant vascular anomaly where a direct connection forms between an artery and a vein, effectively bypassing the capillary bed. In a healthy circulatory system, blood flows from high-pressure arteries into microscopic capillaries, where the exchange of oxygen and nutrients occurs, before draining into low-pressure veins. However, when an arteriovenous fistula is present, the high-pressure arterial blood surges directly into the venous system. This short-circuiting alters local and systemic hemodynamics, often leading to visible physical changes and potential cardiovascular complications. While some fistulas are congenital or the result of trauma, others are surgically created to facilitate life-saving medical treatments like hemodialysis. Understanding the mechanisms, clinical signs, and management of these vascular connections is essential for preventing long-term damage to the heart and surrounding tissues.
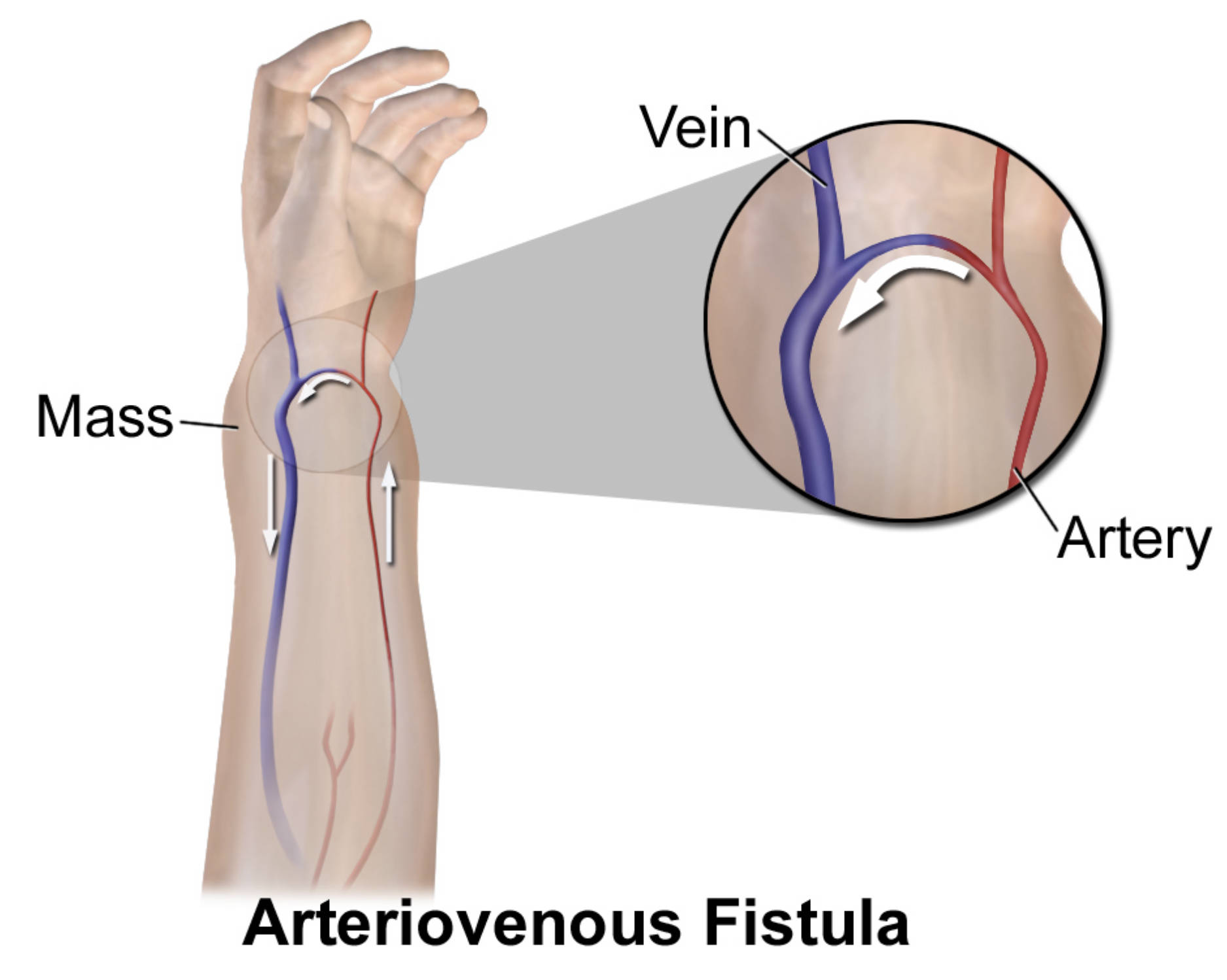
Mass: This term describes the localized swelling or lump that often develops at the site of the fistula due to the high-pressure blood flow distending the vessels. In many patients, this mass may exhibit a palpable vibration known as a thrill or a visible pulsation that matches the heartbeat.
Vein: The vein in this illustration is the vessel receiving redirected arterial blood, causing it to undergo structural changes such as dilation and wall thickening. Over time, the increased pressure causes the vein to become “arterialized,” making it larger and tougher than a standard peripheral vein.
Artery: The artery is the source of the high-pressure blood flow that is being diverted away from its intended capillary network. Because blood follows the path of least resistance, a significant portion of arterial output is channeled through the fistula, which can sometimes lead to reduced blood supply in the tissues further down the limb.
The Complex Hemodynamics of Arteriovenous Connections
The primary physiological impact of a fistula is the drastic change in hemodynamics within the affected region. Because veins are structurally designed to handle low-pressure return flow, the sudden influx of high-pressure arterial blood causes the venous walls to stretch and hypertrophy. This process is often beneficial when a fistula is surgically created for dialysis, as it allows the vein to be punctured repeatedly by large-gauge needles. However, in pathological cases, this pressure can lead to venous hypertension, which manifests as swelling, skin changes, and even ulceration in the extremities.
Systemically, the heart must work significantly harder to maintain adequate blood pressure because the fistula acts as a low-resistance circuit. This increases the total cardiac output, as the heart pumps more blood to compensate for the volume being “lost” through the shunt. If the fistula is large enough and left untreated for a prolonged period, the chronic volume overload can lead to the development of high-output heart failure, where the heart eventually becomes unable to meet the body’s metabolic demands despite pumping a larger-than-normal volume of blood.
Etiology: How Do Arteriovenous Fistulas Form?
Arteriovenous fistulas can be categorized into three main types based on their origin: congenital, acquired, or surgically created. Congenital fistulas are present at birth and result from developmental errors during the formation of the vascular system. These are often complex and may involve multiple small connections rather than a single large channel. Acquired fistulas usually result from penetrating trauma, such as a stab or gunshot wound, where an adjacent artery and vein are injured simultaneously and heal together. They can also occur as a rare complication of medical procedures like cardiac catheterization or biopsies.
- Traumatic Injury: Sharp force trauma that pierces both a neighboring artery and vein is a leading cause of accidental AVFs.
- Erosive Processes: Inflammatory conditions or tumors can occasionally erode the walls of adjacent vessels, creating a spontaneous connection.
- Genetic Factors: Certain conditions like Hereditary Hemorrhagic Telangiectasia (HHT) predispose individuals to multiple vascular malformations.
The Role of Arteriovenous Fistulas in Hemodialysis
In the context of chronic kidney disease, an arteriovenous fistula is often considered the “gold standard” for vascular access. Surgeons intentionally create a connection between an artery (often the radial or brachial artery) and a nearby vein (such as the cephalic vein) in the arm. This surgical AVF requires a “maturation” period of several weeks to months, during which the vein grows larger and its walls thicken in response to the arterial pressure. Once matured, the fistula provides the high flow rates necessary for the dialysis machine to efficiently filter toxins from the blood.
Compared to synthetic grafts or central venous catheters, surgically created fistulas have lower rates of infection and a longer lifespan. However, they are not without risks. Patients must be monitored for “steal syndrome,” where too much blood is diverted away from the hand, leading to pain, coldness, and numbness. Additionally, the high-flow environment can eventually lead to the formation of aneurysms or stenoses within the fistula, requiring interventional maintenance to keep the access point functional for the patient’s ongoing treatment.
Clinical Presentation and Diagnostic Red Flags
Small arteriovenous fistulas may remain asymptomatic for years, but larger ones typically present with unmistakable clinical signs. One of the most classic findings is the Nicoladoni-Branham sign, where the patient’s heart rate slows down immediately when the fistula is compressed manually. Patients often report hearing a continuous “whooshing” sound in the affected limb, which corresponds to the turbulent blood flow across the narrow opening. Physically, the skin over the fistula may feel warmer than the surrounding area due to the increased blood volume.
Other symptoms include visible bulging veins that may resemble varicose veins, but with a distinct pulse. In the legs, a fistula can cause significant edema and skin pigmentation changes that mimic chronic venous insufficiency. If a patient presents with unexplained heart failure symptoms—such as shortness of breath or exercise intolerance—and has a history of vascular trauma or dialysis access, a hidden or high-flow fistula must be ruled out as a contributing factor.
Advanced Diagnostic and Imaging Modalities
The initial diagnosis of an AVF is often made through a physical examination and the use of a stethoscope to listen for a bruit. To confirm the diagnosis and map the anatomy, Duplex Ultrasound is the first-line imaging choice. It allows the clinician to visualize the direct communication between vessels and measure the velocity of the blood flow. High-velocity, low-resistance flow patterns in the supplying artery and pulsatile flow in the draining vein are hallmark sonographic indicators of a fistula.
For more complex cases or for surgical planning, Computed Tomography Angiography (CTA) or Magnetic Resonance Angiography (MRA) provides a three-dimensional view of the vascular tree. However, the definitive “gold standard” remains conventional digital subtraction angiography. This invasive procedure involves injecting contrast dye directly into the arteries under X-ray guidance, allowing the radiologist to see exactly where the blood is shunting. This modality is particularly useful because it can often transition immediately into a therapeutic intervention.
Modern Treatment and Management Strategies
Treatment for an arteriovenous fistula depends on its size, location, and whether it is causing symptoms or complications. Small, asymptomatic fistulas in non-critical areas may simply be monitored with regular ultrasound. For those that require intervention, percutaneous embolization has become a highly effective minimally invasive option. During this procedure, an interventional radiologist uses a catheter to place small coils, plugs, or liquid embolic agents into the fistula to block the abnormal connection and restore normal flow patterns.
In cases where the fistula involves large vessels or is unsuitable for endovascular techniques, traditional surgical repair may be necessary. This involves dissecting the area and either suturing the opening closed or interposing a graft to repair the damaged vessels. For dialysis patients, management focuses on preserving the fistula through “fistuloplasty” (balloon angioplasty) if narrowing occurs. Regardless of the method, the goal of treatment is always to eliminate the abnormal shunt while preserving the blood supply to the distal tissues and reducing the workload on the heart.
Conclusion and Future Perspectives
Arteriovenous fistulas are complex vascular lesions that highlight the body’s incredible ability to adapt to altered pressure environments. While they serve as a lifeline for thousands of dialysis patients, pathological fistulas pose a serious threat to cardiovascular health if left undiagnosed. Advances in imaging and endovascular technology have transformed the management of these conditions, moving away from high-risk open surgeries toward precise, catheter-based interventions. By recognizing the early signs—such as a palpable thrill or an unusual mass—and utilizing modern diagnostic tools, clinicians can effectively manage these anomalies, ensuring both the integrity of the vascular system and the long-term health of the patient’s heart.

