The biological journey of the blood fluke, a parasitic trematode of the genus Schistosoma, is one of the most complex and fascinating sagas in the world of microbiology and tropical medicine. This parasite is responsible for a chronic and debilitating disease that affects millions of people across Africa, Asia, and South America, particularly in regions where clean water and sanitation are scarce. Unlike many other parasites that are ingested, Schistosoma has evolved a highly specialized mechanism to penetrate intact human skin, allowing it to bypass the primary barriers of the host. The cycle involves a mandatory transit through a specific freshwater snail, acting as an essential biological incubator where the parasite multiplies exponentially before seeking out its human host. By understanding the intricacies of this multi-staged life cycle, medical professionals and public health officials can better implement strategies for prevention, diagnosis, and treatment. This article provides an in-depth exploration of how these organisms transition from microscopic eggs to resilient adult worms, highlighting the physiological and environmental factors that sustain their survival.
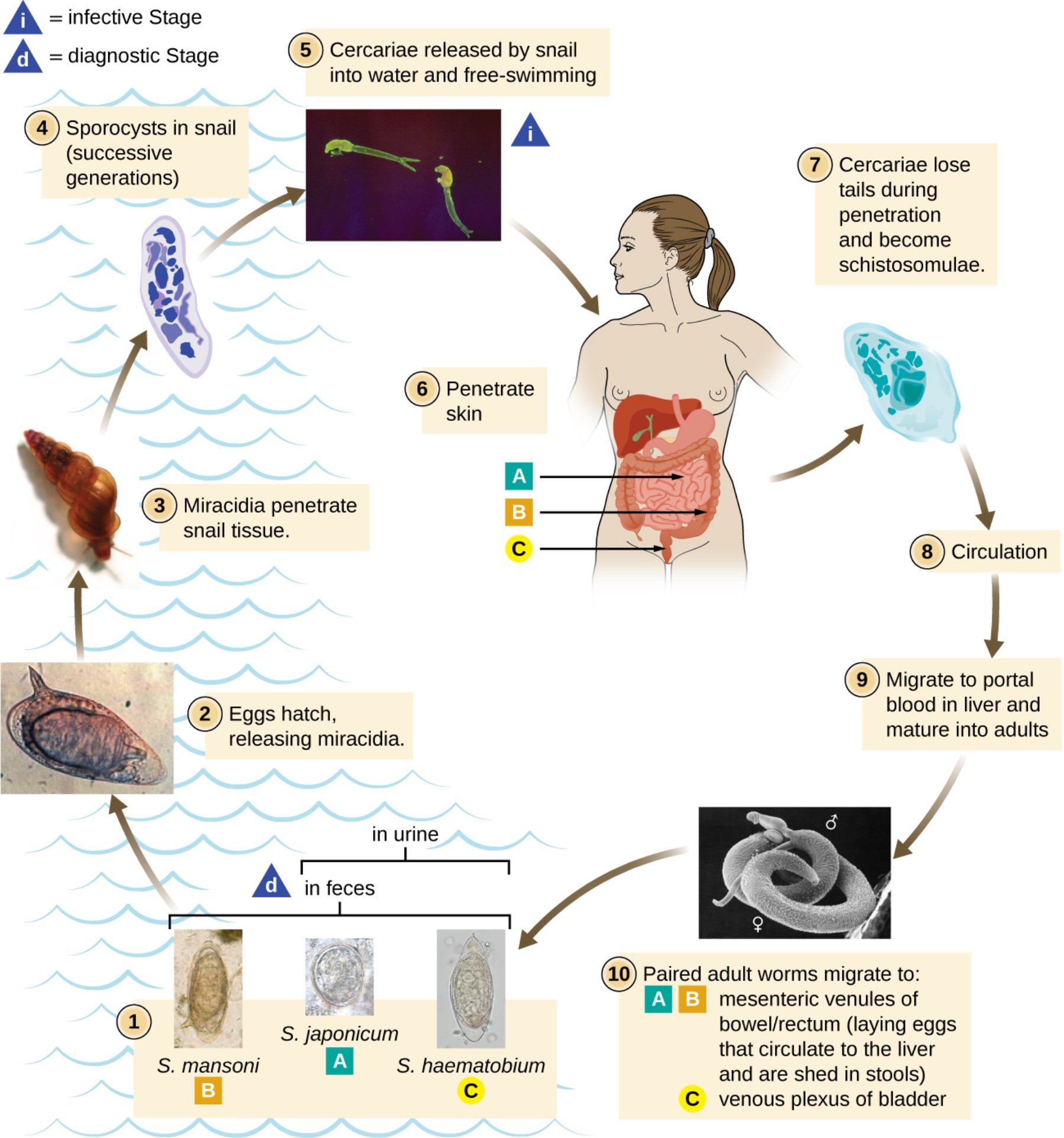
S. mansoni, S. japonicum, and S. haematobium eggs: These are the diagnostic stages of the parasite, characterized by distinct morphological features like the lateral spine in S. mansoni or the terminal spine in S. haematobium. They are shed from the human body into the environment via feces or urine, depending on the specific species of the fluke.
Eggs hatch, releasing miracidia: Once the eggs reach fresh water, they absorb moisture and rupture, releasing a ciliated, free-swimming larval stage known as a miracidium. These organisms are highly motile and use chemical sensors to locate their specific intermediate snail host within a few hours of hatching.
Miracidia penetrate snail tissue: Upon finding a suitable freshwater snail, the miracidium uses specialized enzymes to burrow into the soft tissues of the snail. This marks the transition from the external environment into the first host of the life cycle.
Sporocysts in snail: Inside the snail, the parasite undergoes a remarkable process of asexual reproduction, transforming into mother and then daughter sporocysts. This stage allows a single miracidium to eventually produce thousands of infectious larvae, significantly increasing the parasite’s chances of reaching a human host.
Cercariae released by snail: These are forked-tail, free-swimming larvae that emerge from the snail into the water in response to light or temperature cues. They represent the infective stage, actively seeking out human skin to begin the next phase of the cycle.
Penetrate skin: When a human enters contaminated water, the cercariae attach to the skin and utilize proteolytic enzymes to penetrate the epidermis. This process is rapid and often results in a temporary, itchy rash often referred to as ‘swimmer\’s itch.’
Cercariae lose tails and become schistosomulae: Immediately after entering the human body, the larvae shed their swimming tails to adapt to the internal environment. Now called schistosomulae, they begin to migrate through the dermal layers to reach the circulatory system.
Circulation: The young parasites enter the venous blood flow and are carried through the right side of the heart into the lungs. They eventually pass into the systemic circulation, being distributed to various organs until they reach the liver’s portal system.
Migrate to portal blood in liver: In the vessels of the liver, the schistosomulae mature into adult male and female worms and form permanent pairs. The male worm possesses a long groove called a gynecophoral canal, where the thinner female resides for the duration of their long lifespan.
Paired adult worms migrate to final sites: The coupled pairs move against the blood flow to reach the mesenteric veins of the intestines (S. mansoni and S. japonicum) or the venous plexus of the bladder (S. haematobium). In these final destinations, the females produce hundreds of eggs daily, continuing the cycle as they are excreted by the host.
The Aquatic Stage: The Role of the Intermediate Host
The survival of the Schistosoma parasite is entirely dependent on the presence of fresh water and a specific Intermediate Host. The eggs released from the human body require a freshwater environment to hatch. If the eggs are deposited on dry land, they quickly perish, which is why schistosomiasis is predominantly a disease of riverine and lakeside communities. Once hatched, the miracidium is a race against time; it has approximately 8 to 12 hours to find a snail before its energy reserves are depleted. The specificity of the snail host is remarkable; S. mansoni requires snails of the genus Biomphalaria, while S. haematobium seeks out Bulinus snails.
Inside the snail, the parasite is effectively shielded from the outside world while it multiplies. This asexual phase is a critical bottleneck in the life cycle. The snail provides the necessary nutrients for the sporocysts to grow and divide. If the snail population is reduced through environmental changes or targeted molluscicides, the cycle is effectively broken. This biological vulnerability is a primary target for public health interventions aimed at reducing the prevalence of Schistosomiasis in endemic regions.
The Human Infection: Cercarial Penetration and Migration
The transition from the snail back into a Definitive Host (the human) is mediated by the cercariae. These larvae are equipped with a tail that allows for powerful swimming and a head filled with penetration glands. When a cercaria encounters human skin, it doesn’t just sit on the surface; it utilizes mechanical drilling movements and chemical secretions to dissolve the skin’s barrier. This process is so efficient that the parasite can be completely submerged beneath the skin surface within minutes of contact.
The journey from the skin to the liver is a treacherous one for the schistosomulum. It must navigate the host’s immune system, which often recognizes the foreign proteins on the parasite’s surface. To counter this, the schistosomulum incorporates host proteins into its own outer membrane, effectively “cloaking” itself from immune detection. As it travels through the lungs, it may cause a transient cough or fever. The final destination, the portal venous system of the liver, is the only place where the conditions are perfect for the worms to reach sexual maturity and find their mates.
Pathogenesis: The Damage Caused by Eggs
A common misconception about this disease is that the adult worms cause the primary damage. In reality, the adult worms are remarkably well-tolerated by the human body and can live for decades without causing significant symptoms. The true pathology of the disease is caused by the eggs. While many eggs successfully exit the body through the bowel or bladder, a significant percentage become trapped in the surrounding tissues or are swept back by the blood flow into the liver or other organs.
The host’s immune system reacts violently to these trapped eggs, forming small clusters of inflammatory cells called granulomas. Over time, this chronic inflammation leads to extensive scarring (fibrosis). In the case of intestinal species, this can cause liver cirrhosis and portal hypertension. For S. haematobium, the inflammation occurs in the bladder and ureters, which can lead to hematuria (blood in the urine), kidney failure, and a significantly increased risk of bladder cancer. This egg-induced damage is the hallmark of chronic infection and the main cause of long-term morbidity.
Clinical Presentation and Katayama Fever
The clinical manifestations of infection vary depending on the stage of the cycle and the host’s prior exposure. Shortly after infection, some individuals develop Katayama fever, a systemic hypersensitivity reaction to the migrating larvae and the onset of egg production. Symptoms include fever, chills, cough, and muscle aches. This acute phase is most commonly seen in travelers or people who have moved into an endemic area for the first time, as their immune systems have not yet been desensitized to the parasite’s antigens.
Chronic infection is often more subtle but much more dangerous. Children in endemic areas often suffer from anemia and stunted growth because the constant egg production and immune response drain the body’s resources. In adults, the complications of organ fibrosis dominate the clinical picture. Because the symptoms develop slowly over years, many patients do not seek treatment until irreversible damage has already occurred in the liver or urinary system. This highlights the importance of regular screening programs in at-risk populations.
Diagnosis, Treatment, and Public Health Control
Diagnosis of the disease typically involves the microscopic examination of stool or urine samples to identify the characteristic eggs. Advanced diagnostic techniques also include blood tests to detect antibodies or specific parasite antigens. Once diagnosed, the primary treatment is Praziquantel. This medication is highly effective at killing adult worms of all Schistosoma species. It works by increasing the permeability of the worm’s membranes to calcium, causing paralysis and eventual death. While the drug is effective at clearing the infection, it cannot reverse the extensive scarring that may have already formed in the organs.
Controlling the spread of the parasite requires a multi-faceted approach. Large-scale “mass drug administration” (MDA) programs aim to treat entire communities simultaneously to reduce the total number of eggs being shed into the environment. However, medication alone is rarely enough for permanent elimination. Lasting success requires improvements in sanitation to prevent feces and urine from reaching water bodies, as well as the provision of safe water for bathing and washing. Education also plays a vital role, teaching people to avoid high-risk water areas during the peak times of cercarial release.
Conclusion: Breaking the Cycle
The life cycle of Schistosoma is a testament to the incredible adaptability of parasites. By utilizing both a mollusk and a mammal to complete its development, the fluke ensures its continued survival across diverse environments. However, this complex cycle also provides multiple points of intervention. Whether it is through the elimination of the snail host, the chemical treatment of human patients with medications like Praziquantel, or the fundamental improvement of global sanitation, we have the tools to disrupt this cycle. Eradicating schistosomiasis remains one of the great challenges of modern global health, but with continued investment and a deep understanding of the parasite’s biology, it is a goal that remains within our reach. By focusing on the intersection of human behavior, environmental health, and medical science, we can protect the millions of people who currently live at risk of this ancient and persistent disease.
Schistosoma, Life Cycle, Miracidia, Cercariae, Schistosomiasis, Snail Host, Microbiology, Parasitology, Praziquantel, Tropical Medicine

