In the realm of human physiology, maintaining a stable internal environment is paramount for cellular survival. This stability is largely governed by the principles of osmosis and tonicity, which dictate how water moves across semi-permeable membranes. The featured image provides a microscopic visualization of a red blood cell (RBC) suspended in an isotonic medium. In this state, the concentration of non-penetrating solutes outside the cell is equal to that inside the cell, ensuring that the cell preserves its structural integrity and functional capacity. Understanding these dynamics is fundamental for medical practitioners, particularly when managing fluid resuscitation and electrolyte balance in clinical settings.
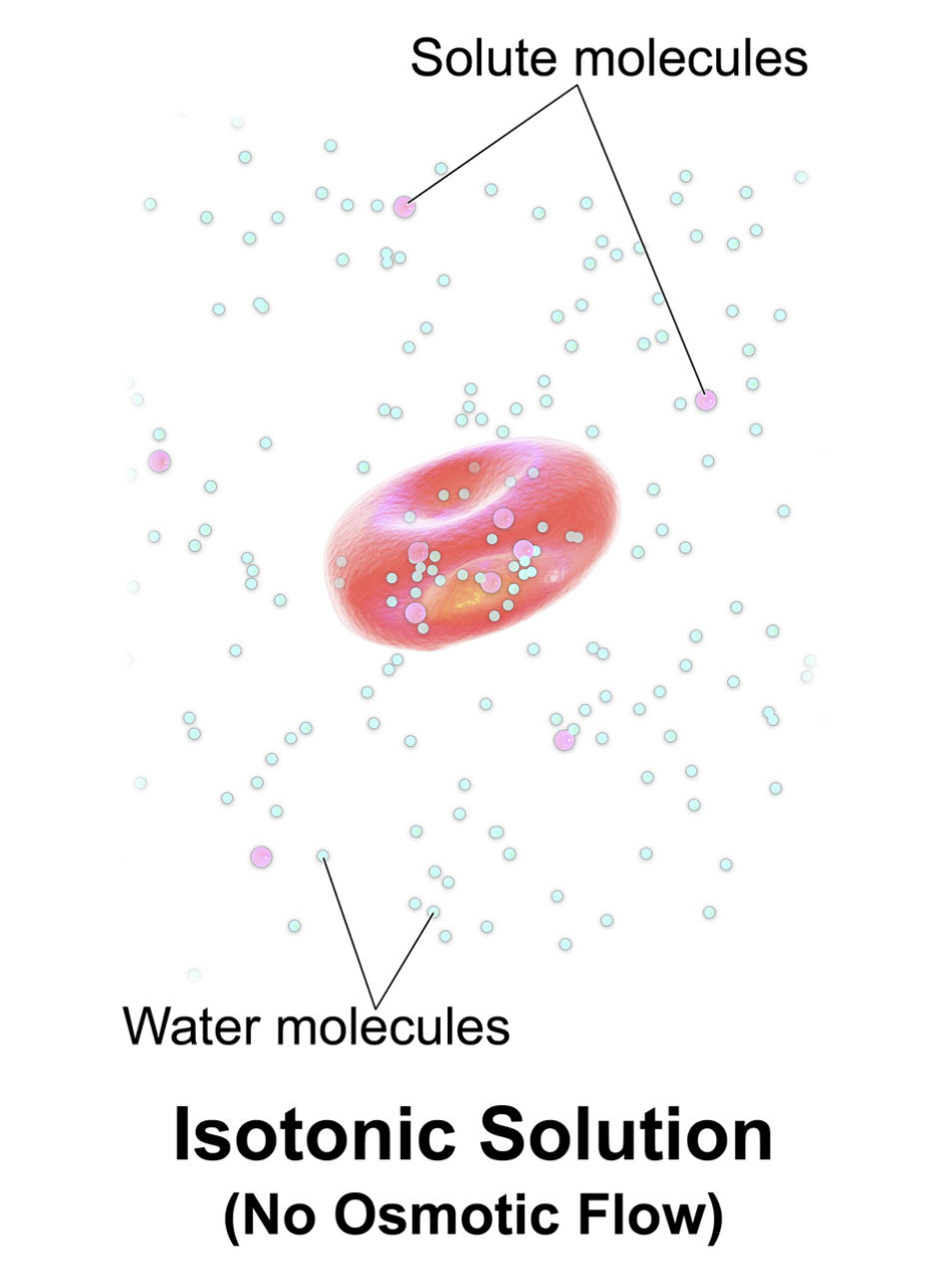
Label-by-Label Explanation
Solute molecules: These represent particles, such as ions or glucose, dissolved in the solvent. In a physiological context, these solutes are often restricted from freely crossing the cell membrane, thereby creating the osmotic pressure necessary to hold water within a specific compartment.
Water molecules: These are the primary solvent molecules that move through the cell membrane via simple diffusion or specialized channels called aquaporins. Their movement is driven by the concentration gradient of solutes, moving from areas of low solute concentration to areas of high solute concentration.
Isotonic Solution (No Osmotic Flow): This describes a state of dynamic equilibrium where the osmolality of the extracellular fluid matches that of the intracellular fluid. While water molecules continue to move back and forth across the membrane, the net movement is zero, which allows the red blood cell to maintain its characteristic biconcave disc shape.
Physiological Significance of Tonicity
Tonicity refers to the ability of an extracellular solution to make water move into or out of a cell by osmosis. It is specifically related to the concentration of non-penetrating solutes—those that cannot cross the cell membrane. In the image, we see the ideal physiological state for a red blood cell. Because the solution is isotonic, the cell is neither bloated with excess water nor shriveled from water loss.
The red blood cell’s biconcave shape is not merely an aesthetic feature; it is a functional necessity. This geometry provides a high surface-area-to-volume ratio, which optimizes the diffusion of oxygen and carbon dioxide across the membrane. If the cell were to become spherical in a hypotonic solution or shrunken (crenated) in a hypertonic solution, its ability to navigate narrow capillaries and exchange gases efficiently would be significantly compromised.
Clinical Relevance and Fluid Therapy
In clinical practice, the concept of an isotonic solution is most frequently encountered during the administration of intravenous (IV) fluids. When a patient is dehydrated or has lost blood, healthcare providers must restore volume without causing cellular damage. The most common isotonic solutions used in hospitals include:
- 0.9% Normal Saline (NS): A solution of sodium chloride that closely mimics the sodium concentration of plasma.
- Lactated Ringer’s (LR): A balanced crystalloid containing sodium, chloride, potassium, calcium, and lactate, often used for surgical patients or burn victims.
- 5% Dextrose in Water (D5W): While technically isotonic in the bag, it becomes hypotonic once the body metabolizes the glucose, making its clinical application distinct from NS or LR.
Administering an incorrect fluid can have dire consequences. For instance, giving a severely dehydrated patient a purely hypotonic solution too rapidly can lead to cerebral edema as water rushes into brain cells. Conversely, hypertonic solutions are used sparingly to treat specific conditions like symptomatic hyponatremia, as they pull water out of cells into the vascular space.
The Difference Between Osmolarity and Tonicity
A common point of confusion for medical students is the distinction between osmolarity and tonicity. Osmolarity is a chemical measurement based on the total number of solute particles per liter of solution, regardless of whether those particles can cross the membrane. Tonicity, however, is a biological concept that only considers the concentration of solutes that do not cross the membrane.
For example, urea is a penetrating solute. A solution of urea might have the same osmolarity as plasma (iso-osmotic), but because urea can move freely into the cell, water will follow it, causing the cell to swell. Therefore, a solution can be iso-osmotic but still be functionally hypotonic. The image provided illustrates a true isotonic state where both osmolarity and the effective osmotic pressure (tonicity) are balanced.
Key Learning Points for Medical Learners
When analyzing cellular environments, always distinguish between the chemical concentration of a solution and its physical effect on the cell. The lipid bilayer acts as a gatekeeper, and the behavior of the cell as shown in the diagram—maintaining a healthy biconcave shape—is the benchmark for successful fluid homeostasis. Remember that isotonicity is a comparative term; it describes the relationship between the inside and the outside of the cell. In practice, maintaining this balance is the primary goal of maintenance fluid therapy to prevent complications like hemolysis or tissue ischemia.
Medical Learning Tips
- Tonicity is determined only by solutes that cannot cross the semi-permeable cell membrane.
- In an isotonic solution, the net movement of water is zero, though molecules move in both directions at equal rates.
- The biconcave shape of the red blood cell is preserved in isotonic environments to maximize gas exchange efficiency.

