The centrosome serves as the primary microtubule-organizing center in animal cells, coordinating the assembly of microtubules essential for cell division, intracellular transport, and maintaining cell shape. Composed of two centrioles surrounded by pericentriolar material, it plays a pivotal role during mitosis by forming the poles of the mitotic spindle.
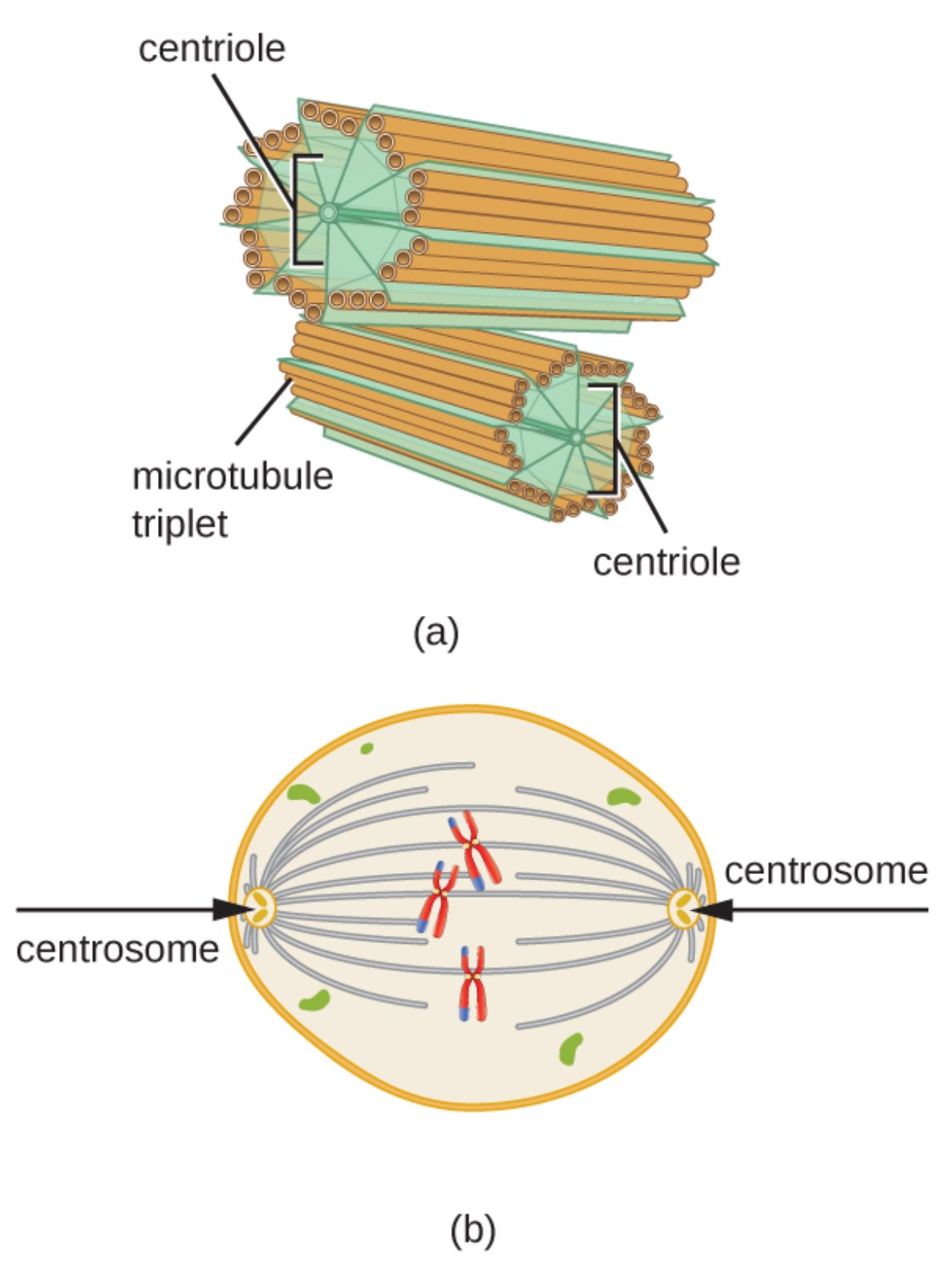
Key Labeled Elements in the Centrosome Diagram
Centriole Centrioles are cylindrical structures that form the core of the centrosome, typically appearing in pairs oriented at right angles to each other. Each centriole consists of nine triplet microtubules arranged in a characteristic 9+0 pattern, providing structural stability and serving as a template for basal bodies in cilia and flagella formation.
Microtubule triplet Microtubule triplets are groups of three microtubules fused together, making up the wall of each centriole. These triplets, composed of alpha and beta tubulin dimers with associated proteins like gamma-tubulin, contribute to the rigidity of centrioles and enable the nucleation of new microtubules from the pericentriolar material.
Centrosome The centrosome is the main microtubule-organizing center (MTOC) in animal cells, encompassing two centrioles and a cloud of pericentriolar material rich in gamma-tubulin ring complexes. It orchestrates microtubule polymerization, ensuring proper spindle formation during cell division and facilitating polarized cell functions.
The centrosome is a critical organelle in animal cells, acting as the hub for microtubule organization and duplication. Its structure features two barrel-shaped centrioles, each built from nine microtubule triplets, positioned perpendicularly to prevent interference during microtubule nucleation. This orthogonal arrangement allows the mature mother centriole to anchor microtubules effectively while the daughter centriole matures over cell cycles.
During interphase, the centrosome maintains cellular architecture by radiating microtubules that support transport and polarity. As cells enter mitosis, the centrosome duplicates, and each new centrosome migrates to opposite poles, nucleating astral, kinetochore, and polar microtubules to form the mitotic spindle. This bipolar structure ensures accurate chromosome alignment and segregation, preventing errors that could lead to aneuploidy.
Key roles of the centrosome include:
- Organizing the microtubule cytoskeleton for cell shape and motility
- Serving as the basal body for cilia and flagella in specialized cells
- Directing spindle pole formation during mitosis
- Coordinating cytokinesis for proper cell division
From a medical standpoint, centrosome abnormalities are frequently observed in cancer cells, where supernumerary centrosomes lead to multipolar spindles and chromosomal instability, a hallmark of malignancy. This clustering of extra centrosomes allows cancer cells to divide bipolarly despite defects, promoting tumor progression. Targeted therapies aiming at centrosome declustering are under investigation to selectively induce lethal multipolar division in cancer cells while sparing normal cells.
Additionally, mutations in centriolar proteins can cause ciliopathies, a group of disorders including primary ciliary dyskinesia and Meckel-Gruber syndrome, characterized by defective cilia function leading to respiratory issues, situs inversus, and developmental anomalies. Understanding centrosome biology continues to inform diagnostics and treatments for these conditions.
In essence, the centrosome’s precise structure and regulatory functions are fundamental to orderly cell division and tissue homeostasis. Disruptions in its activity underscore its relevance in both developmental disorders and oncogenesis, highlighting the need for ongoing research into microtubule-organizing centers for advanced therapeutic strategies.

