Dictyostelium discoideum, a fascinating soil-dwelling amoeba, serves as one of the most powerful model organisms in modern biomedical research. This cellular slime mold transitions from solitary amoeboid cells to a multicellular “slug” and eventually a fruiting body under nutrient stress, revealing fundamental principles of cell motility, chemotaxis, differentiation, and cooperation that mirror processes in human cells. Its genetic tractability, rapid life cycle, and evolutionary position in the Amoebozoa supergroup make it invaluable for studying host-pathogen interactions, neurodegenerative diseases, and basic cellular mechanisms relevant to human health.
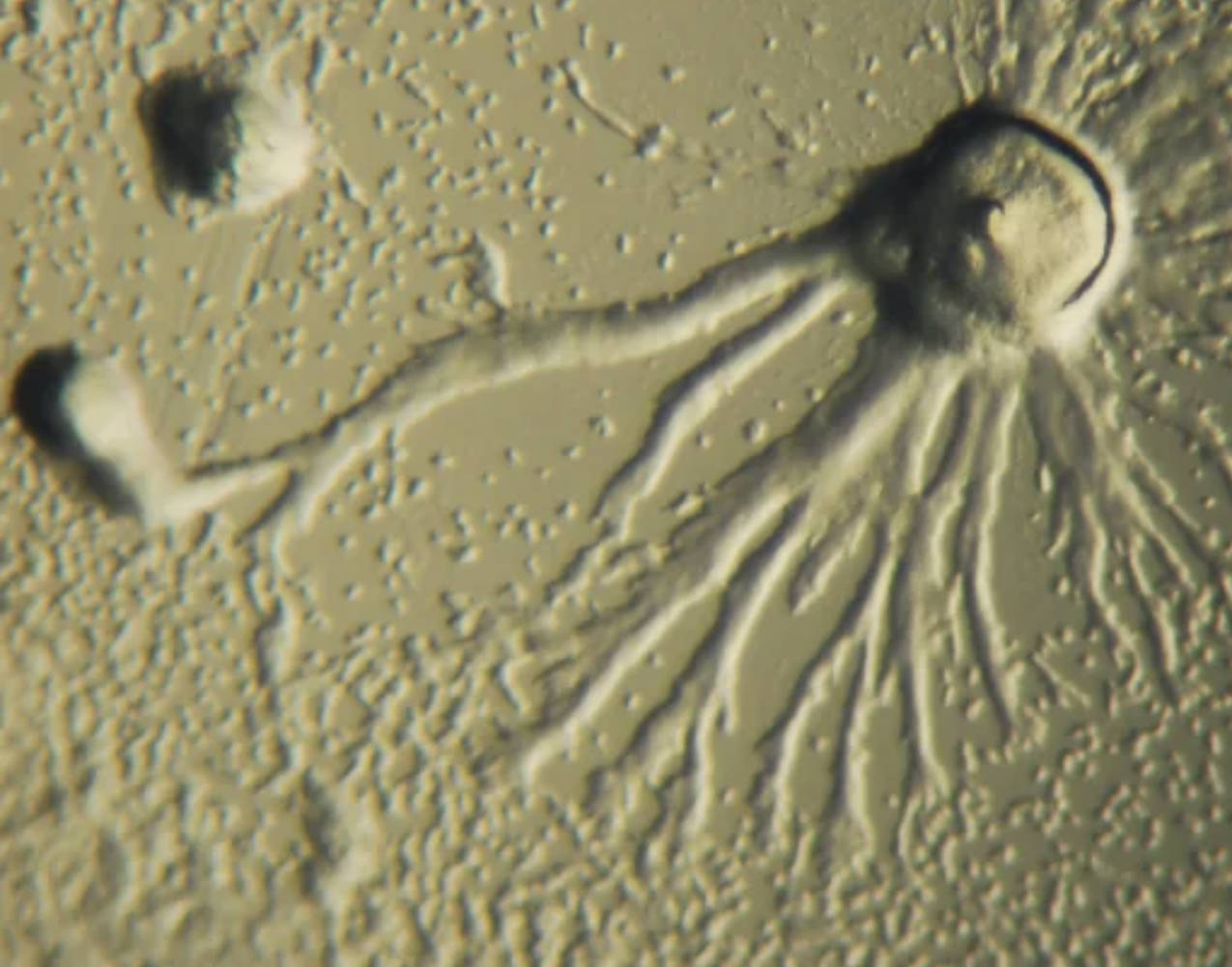
Dictyostelium discoideum is a species of cellular slime mold belonging to the Amoebozoa supergroup. In its vegetative stage, it exists as independent amoeboid cells that feed on bacteria by phagocytosis. Under starvation, thousands of cells aggregate using cyclic AMP as a signal, forming a migratory multicellular structure known as a slug that demonstrates collective behavior and differentiation.
Slug refers to the migratory pseudoplasmodium stage of Dictyostelium discoideum, consisting of up to 100,000 cells moving as a unified body. This structure exhibits directional migration toward light, heat, and humidity while leaving a slime trail, showcasing sophisticated cell-cell communication and polarity essential for understanding developmental biology.
Pseudopodia are the temporary cytoplasmic extensions visible in the image that enable amoeboid movement and feeding. In Dictyostelium, these lobe-like or filopodial projections facilitate chemotactic navigation and phagocytosis, processes conserved across eukaryotes and critical for immune cell function in humans.
Amoeboid cell describes the individual haploid cells of Dictyostelium discoideum during the growth phase. These cells are highly motile, using actin cytoskeleton dynamics to crawl across surfaces, making the organism an excellent model for investigating cytoskeletal disorders and cell migration in cancer or wound healing.
The Life Cycle of Dictyostelium discoideum
Dictyostelium discoideum has a remarkable biphasic life cycle that alternates between unicellular and multicellular phases. Solitary amoebae proliferate by binary fission when bacteria are abundant. Upon nutrient depletion, cells secrete cyclic AMP, attracting neighbors to form streams that converge into a mound, which then elongates into the migratory slug. The slug eventually culminates into a fruiting body with a stalk of dead cells supporting a sorus of dormant spores, completing the cycle.
Why Dictyostelium discoideum Is a Premier Biomedical Model
Recognized by the National Institutes of Health as a key non-mammalian model organism, Dictyostelium discoideum offers unique advantages for medical research. Its haploid genome simplifies genetic manipulations, including gene knockouts and multiple deletions. Cells grow rapidly at room temperature in inexpensive media, reaching high densities without complex equipment. These features allow cost-effective, high-throughput studies of conserved eukaryotic processes.
- Short generation time enables rapid screening of developmental phenotypes.
- Genetic tools support creation of mutant strains for functional analysis.
- Transparency and accessibility facilitate live-cell imaging of dynamic processes.
The organism’s genome encodes homologs of many human disease-related genes, bridging basic cell biology with clinical applications.
Applications in Studying Cell Motility and Chemotaxis
The visible pseudopodia and migratory behavior in the image highlight Dictyostelium’s value for chemotaxis research. Cells detect gradients of cyclic AMP with surface receptors, activating intracellular signaling cascades involving G-proteins, PI3K, and actin polymerization. These pathways are highly conserved with those in human neutrophils and other immune cells, providing insights into inflammation, immune response, and metastatic cancer cell migration.
Host-Pathogen Interactions and Innate Immunity
As a soil amoeba, Dictyostelium discoideum naturally encounters bacteria and other microbes, making it an ideal model for host-pathogen studies. It uses phagocytosis and antimicrobial mechanisms similar to macrophages. Researchers use it to investigate bacterial virulence factors, intracellular survival strategies of pathogens like Legionella and Mycobacterium, and the evolution of innate immunity. Findings often translate to understanding human infections and immune deficiencies.
Dictyostelium as a Model for Neurodegenerative Diseases
Recent studies leverage Dictyostelium discoideum to explore mechanisms underlying Alzheimer’s, Parkinson’s, Huntington’s, and other neurological disorders. The organism expresses homologs of human proteins involved in protein aggregation, autophagy, and mitochondrial function. Its simple multicellular development allows rapid assessment of how disease-associated mutations disrupt cell differentiation, motility, or survival. Pharmacological screening in this system helps identify potential therapeutic compounds targeting conserved pathways.
Research Techniques and Imaging in Dictyostelium Studies
Microscopy techniques, such as the phase-contrast or differential interference contrast view shown, reveal fine details of pseudopodia dynamics and slug morphology. Modern approaches include fluorescent tagging of proteins, live-cell time-lapse imaging, and microfluidics for precise chemotactic gradient control. These tools enable quantitative analysis of cellular behaviors, contributing to discoveries in cytoskeletal regulation and signal transduction.
Broader Impacts on Developmental and Evolutionary Biology
Beyond medicine, Dictyostelium discoideum illuminates the origins of multicellularity. The transition from solitary cells to cooperative structures raises questions about altruism, cheating, and kin selection. Comparative genomics with other amoebozoa and opisthokonts refines our understanding of eukaryotic evolution, including the Amoebozoa supergroup’s position relative to animals and fungi.
Practical Considerations for Laboratory Use
Dictyostelium discoideum is straightforward to culture on bacterial lawns or axenic media. Strains are available from stock centers, with extensive genomic resources at dictyBase. Researchers can easily generate mutants and perform complementation assays. Its safety profile allows use in standard laboratory settings without special containment, facilitating widespread adoption in educational and research institutions.
Future Directions in Dictyostelium Research
Advances in CRISPR-based editing, single-cell transcriptomics, and high-resolution imaging promise deeper insights into cellular decision-making. Integrating Dictyostelium models with human iPSC-derived cells or organoids will strengthen translational relevance. As antibiotic resistance and neurodegenerative diseases rise, this humble amoeba continues offering innovative solutions through its conserved biology and experimental accessibility.
Conclusion: The Enduring Value of a Simple Model Organism
From the intricate pseudopodia and collective slug migration captured in the image, Dictyostelium discoideum exemplifies how studying simple eukaryotes yields profound insights into complex human biology. Its contributions span cell biology, immunology, neurobiology, and drug discovery, underscoring the importance of diverse model systems in advancing medical science. Continued research with this organism will undoubtedly uncover new mechanisms and therapeutic avenues for improving human health.

