Osmosis is a fundamental biological process that governs the movement of water across semi-permeable membranes, maintaining the delicate balance of fluids within and around our cells. In the human body, perhaps no cell is more sensitive to these fluid shifts than the erythrocyte, or red blood cell. Lacking a nucleus and possessing a highly specialized protein-lipid bilayer, the red blood cell acts as a biological osmometer. When the concentration of solutes in the surrounding plasma changes, the cell must react instantly to prevent either shriveling or bursting. This phenomenon is not merely a classroom experiment but a critical factor in clinical medicine, influencing how healthcare providers administer intravenous fluids and how the body manages internal hydration. Understanding the relationship between solute concentration and cellular volume is key to grasping the mechanics of human physiology and the life-sustaining nature of fluid homeostasis.
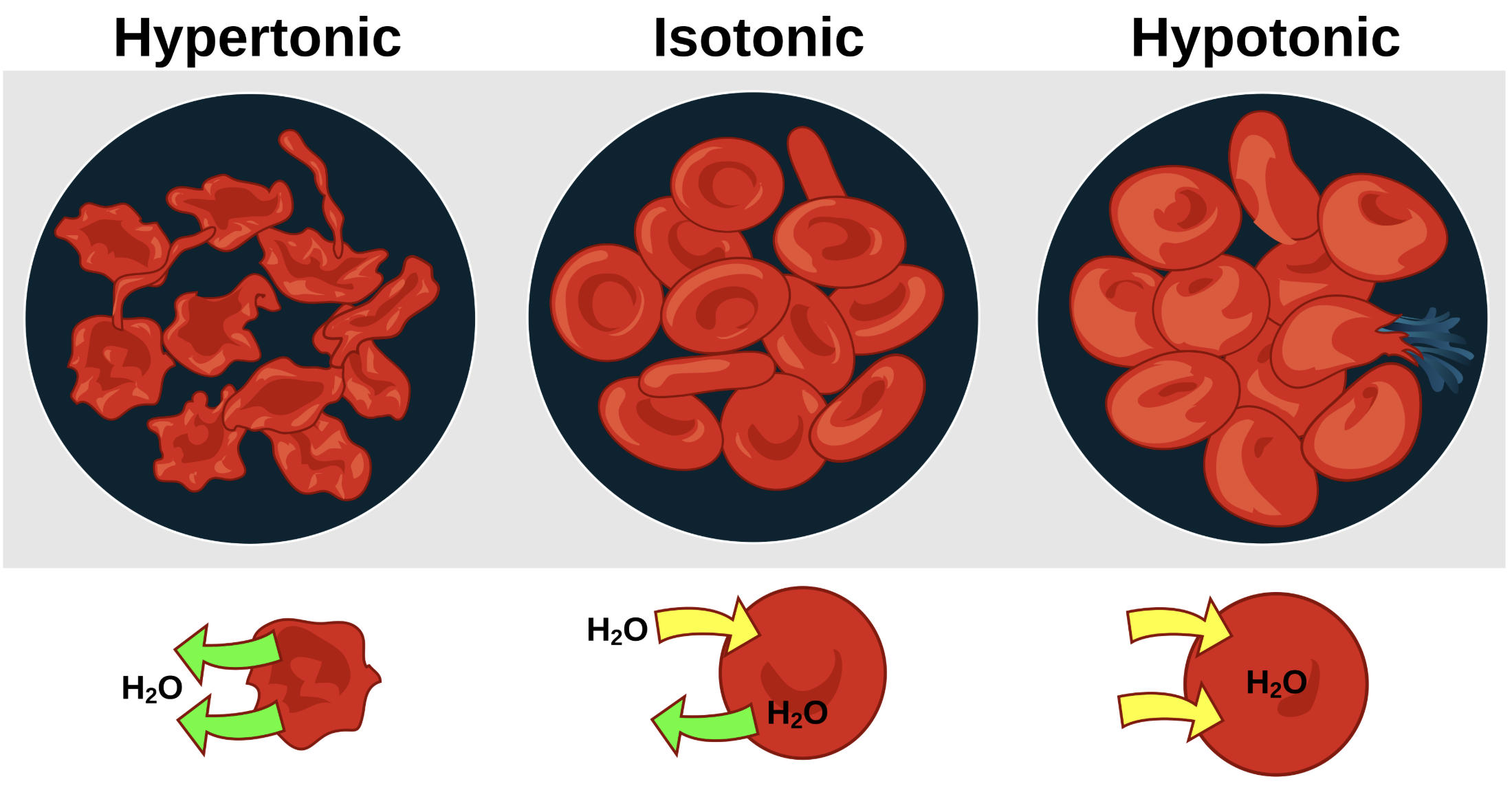
Hypertonic: This term describes an environment where the external solution has a higher concentration of non-permeable solutes than the cell cytoplasm, drawing water out of the cell. As water exits to equalize the concentration gradient, the red blood cell undergoes a process called crenation, resulting in a shriveled and distorted appearance.
Isotonic: In an isotonic state, the solute concentration in the extracellular fluid is exactly equal to that inside the red blood cell, leading to no net movement of water. This is the physiological ideal, allowing the cell to maintain its characteristic biconcave disc shape which is optimized for oxygen and carbon dioxide transport.
Hypotonic: A hypotonic solution contains a lower concentration of solutes relative to the cell’s interior, creating a pressure gradient that forces water into the erythrocyte. The cell swells significantly under this pressure and can eventually reach its breaking point, leading to hemolysis, where the membrane ruptures and releases hemoglobin into the plasma.
H2O: This represents water molecules, which move across the cell membrane via osmosis in response to solute imbalances. The green arrows indicate the net direction of water travel, showing it leaving the cell in hypertonic conditions, moving equally in isotonic conditions, and entering the cell in hypotonic conditions.
The Mechanics of Osmosis and Membrane Permeability
To understand the effect of fluid shifts on red blood cells, one must first look at the physics of the cell membrane. The plasma membrane of a red blood cell is semi-permeable, meaning it allows small molecules like water to pass through freely while acting as a barrier to larger ions and proteins. This movement of water is driven by osmotic pressure, which can be thought of as the force required to prevent water from moving from an area of low solute concentration to an area of high solute concentration. Inside the cell, proteins like hemoglobin and various ions create a specific internal pressure. When the external environment deviates from this pressure, the laws of thermodynamics dictate that water must move to attempt to reach an equilibrium.
This process is largely passive and occurs through specialized water channels called aquaporins, as well as directly through the lipid bilayer. Because the red blood cell has a very high surface-area-to-volume ratio due to its biconcave shape, it can accommodate some degree of swelling. However, unlike plant cells which have a rigid cell wall to provide structural resistance, animal cells are entirely dependent on maintaining a balanced environment to prevent physical destruction. This dependency makes the regulation of plasma osmolarity one of the most strictly controlled systems in the human body.
Hypertonic Environments and the Danger of Dehydration
When red blood cells are placed in a hypertonic solution, such as a high-salt environment, the osmotic gradient pulls water out of the cell’s cytoplasm. This loss of intracellular volume causes the cell membrane to collapse inward upon itself, creating a spiked or “scalloped” edge in a process known as crenation. This is not just a change in appearance; it is a significant functional impairment. A shriveled red blood cell has a reduced surface area for gas exchange and becomes rigid, making it difficult for the cell to navigate the narrow, winding pathways of the microscopic capillaries.
Clinically, hypertonic states can occur during severe dehydration or in certain metabolic emergencies like hypernatremia (high blood sodium levels). If the plasma becomes too concentrated, the systemic shriveling of red blood cells can impede microcirculation and reduce oxygen delivery to vital tissues. Medical treatments for such conditions must be handled with extreme care, as rehydrating the plasma too quickly can swing the environment from one extreme to another, potentially causing the cells to swell and rupture before they can stabilize.
Isotonic Balance: The Foundation of Homeostasis
Maintaining homeostasis is the primary goal of the body’s renal and endocrine systems. In a healthy individual, the kidneys constantly filter the blood and adjust the excretion of water and salts to ensure that the plasma remains isotonic relative to the interior of the blood cells. The standard reference for an isotonic solution in humans is 0.9% Sodium Chloride (normal saline). When fluids of this concentration are administered intravenously, they expand the blood volume without disturbing the physical integrity of the red blood cells themselves.
The biconcave shape maintained in an isotonic environment is essential for the erythrocyte’s primary function. This shape provides the maximum amount of membrane surface area for oxygen to diffuse into the cell and bind with hemoglobin. Furthermore, the flexibility afforded by this balanced state allows the cell to fold and twist as it passes through vessels that are sometimes smaller in diameter than the cell itself. Without a perfectly tuned isotonic environment, the circulatory system would face frequent mechanical bottlenecks.
Hypotonicity and the Risk of Hemolysis
The opposite extreme occurs in a hypotonic environment, where the external fluid is “dilute” compared to the cell’s contents. In this scenario, water rushes into the cell, causing it to lose its biconcave indentation and become spherical. This increases the tension on the cell membrane significantly. Because the lipid bilayer can only stretch so much, a point is reached where the structural proteins of the cytoskeleton give way. This results in the bursting of the cell, a catastrophic event known as hemolysis.
This is the primary reason why pure, distilled water can never be injected directly into a patient’s bloodstream. The sudden drop in plasma tonicity would cause massive, localized cell death as erythrocytes literally explode upon contact with the water. In laboratory settings, this vulnerability is used to diagnostic advantage in the “osmotic fragility test,” which measures how much a patient’s red blood cells can swell before breaking. Patients with conditions like hereditary spherocytosis have cells that are already nearly spherical and thus burst much more easily than healthy cells, providing a clear indicator of the underlying genetic defect.
The Clinical Application of Osmotic Principles
The principles illustrated in the diagram are applied every day in hospitals across the world. When a patient is severely dehydrated, doctors must choose between isotonic, hypotonic, or hypertonic IV fluids based on the specific electrolyte needs of the patient. For example, if a patient has cerebral edema (swelling of the brain), a hypertonic solution may be used strategically to draw excess water out of the brain tissue and into the bloodstream, where it can be filtered out by the kidneys. Conversely, someone with intracellular dehydration might require a slightly hypotonic solution to encourage water to move into the cells.
Every medical intervention involving fluids is a calculated move to manipulate the body’s osmotic balance. By visualizing how red blood cells react to these different pressures, we can better appreciate the complex biological engineering that keeps our blood flowing. Whether it is the kidneys managing salt levels or a nurse hanging a bag of saline, the ultimate goal is the same: to protect the structural and functional integrity of the red blood cell membrane from the powerful, invisible force of moving water.
Conclusion
The relationship between a cell and its environment is a dynamic struggle for equilibrium. As seen in the effects of osmotic pressure, the red blood cell is a delicate entity that relies entirely on the precise concentration of its surrounding fluid to function. From the distorted spikes of a crenated cell in a hypertonic solution to the tragic rupture of a cell in a hypotonic one, we see the physical consequences of chemical imbalances. Mastering these concepts is essential for anyone in the medical field, as it forms the basis of fluid therapy, renal physiology, and the diagnosis of numerous hematologic disorders. The simple movement of water, directed by osmotic pressure, remains one of the most powerful and essential forces in human life.

