The survival of a cell hinges on its ability to meticulously regulate the entry and exit of molecules. The plasma membrane, with its hydrophobic core, presents a significant barrier to most biological molecules that are polar, large, or electrically charged. While small nonpolar molecules like oxygen can slip through the lipid bilayer unnoticed, vital nutrients like glucose and essential ions like sodium require a more sophisticated method of entry. This is where facilitated diffusion becomes indispensable. As a specific form of passive transport, it allows the cell to acquire necessary materials efficiently without the expenditure of metabolic energy. By utilizing specialized proteins as “gateways,” the cell maintains its internal equilibrium while responding to the fluctuating concentrations of the external environment.
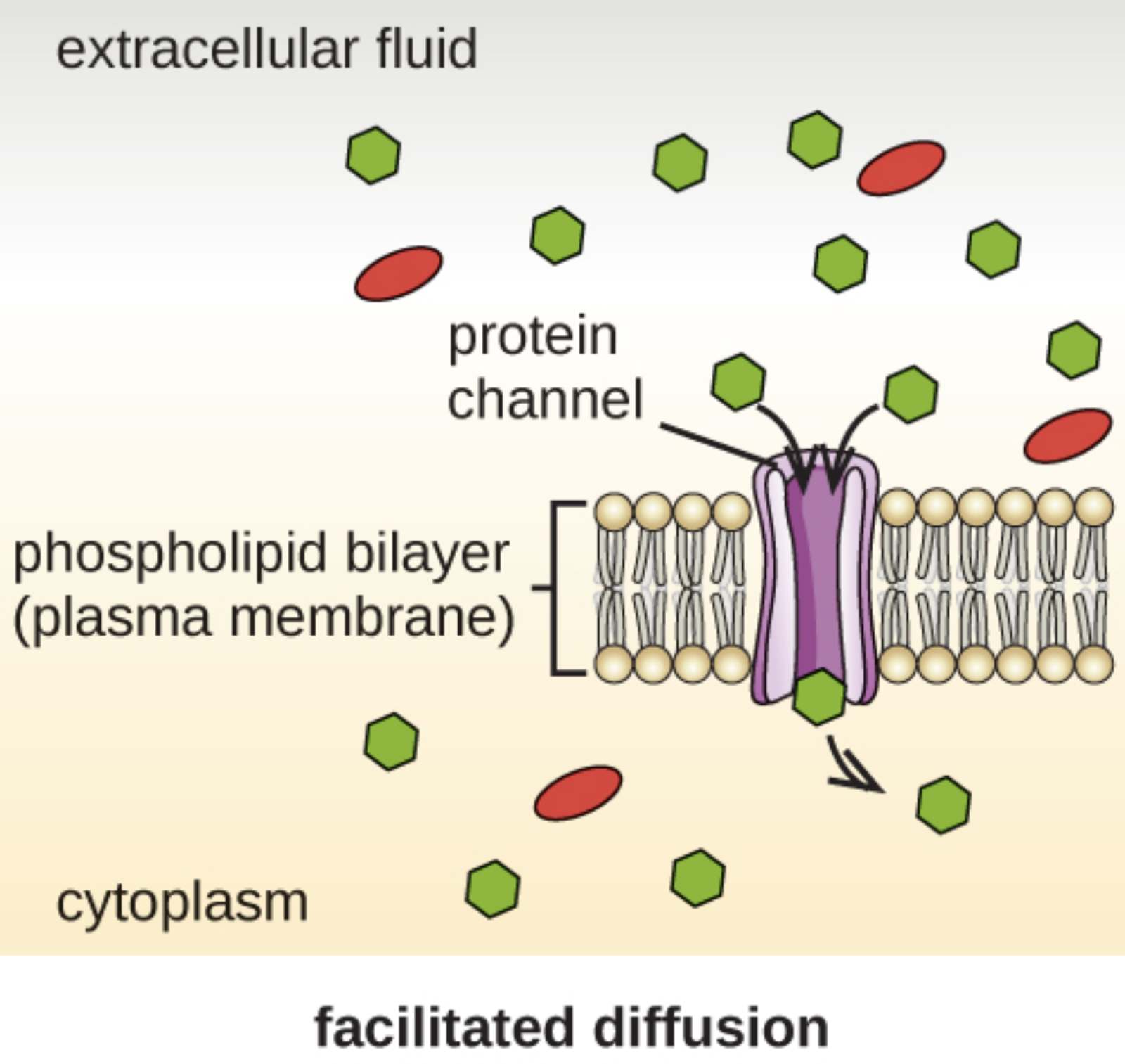
extracellular fluid: This is the aqueous region located outside of the cell’s plasma membrane, containing various nutrients, ions, and signaling molecules. In this diagram, it serves as the area of high concentration from which solutes are moved into the cell.
protein channel: These are integral membrane proteins that form a hydrophilic pore across the phospholipid bilayer. They are highly selective, allowing specific molecules or ions to bypass the hydrophobic lipid interior through a sheltered passage.
phospholipid bilayer (plasma membrane): This dual layer of lipids forms the primary structural boundary of the cell, separating the internal cytoplasm from the external environment. Its hydrophobic center prevents the uncontrolled passage of water-soluble substances, necessitating specialized transport mechanisms.
cytoplasm: This refers to the interior substance of the cell where various organelles are suspended and biochemical reactions take place. Molecules passing through the protein channels are delivered here to be utilized by the cell’s metabolic machinery.
facilitated diffusion: This term describes the passive movement of substances across a biological membrane through specialized transport proteins. This process occurs down a concentration gradient and is essential for the transport of molecules that cannot pass through the lipid bilayer on their own.
The Fundamental Mechanism of Facilitated Diffusion
Facilitated diffusion is a cornerstone concept in passive transport. It operates entirely on the principle of a concentration gradient, meaning molecules move naturally from an area where they are more numerous to an area where they are less numerous. Unlike simple diffusion, which is limited by the physical properties of the lipids themselves, facilitated diffusion is limited by the availability and efficiency of transport proteins embedded in the membrane. This process is categorized as “facilitated” because it requires assistance from the membrane proteins, but it remains a passive endeavor because the driving force is the random thermal motion of the particles, rather than the consumption of cellular ATP.
The selectivity of this process is one of its most remarkable features. Each protein channel or carrier is typically designed to move only one specific type of molecule or a very narrow group of similar substances. This specificity ensures that the cell can precisely control its internal chemistry, allowing potassium ions in while keeping sodium out, or prioritizing the uptake of glucose even when other sugars are present in higher concentrations. This precision is vital for maintaining the resting membrane potential of neurons and the proper osmotic pressure within human tissues.
Types of Transport Proteins: Channels and Carriers
There are two primary categories of proteins involved in this process: channel proteins and carrier proteins. Channel proteins, as highlighted in the diagram, act much like open tunnels. Some are constantly open, acting as leak channels, while others are “gated,” opening only in response to a specific signal. These signals can be chemical ligands, mechanical stress, or changes in electrical voltage. These gated channels are particularly important in the nervous system, where the rapid movement of ions across membranes is necessary for the propagation of nerve impulses.
Carrier proteins, on the other hand, operate with a distinct mechanical action. They bind to the target molecule on one side of the membrane, which triggers a change in the protein’s three-dimensional shape. This conformational change physically carries the molecule across the membrane and releases it on the other side. A classic example is the glucose transporter (GLUT) family. These proteins help move glucose into red blood cells and muscle tissues to fuel cellular respiration, ensuring the body has a steady supply of energy for both basic metabolism and intense physical activity.
Factors Influencing Transport Rate and Saturation
Unlike simple diffusion, where the rate of transport increases linearly as the concentration gradient becomes steeper, facilitated diffusion exhibits a phenomenon known as saturation. Because there are a finite number of channel proteins in any given area of the membrane, the rate of transport can only increase until every protein is working at its maximum capacity. This state is known as Vmax. Once every available protein is occupied by a solute, adding more substance to the extracellular fluid will not result in a faster rate of entry. This is a critical limiting factor in biological systems.
Temperature and the number of proteins also play significant roles. Higher thermal energy generally increases molecular collisions, but because this process relies on protein function, extreme temperatures or changes in pH can denature the transport proteins, effectively shutting down the transport mechanism. Furthermore, the number of proteins in the membrane is not static; cells can often regulate their membrane permeability by inserting more transport proteins into the membrane or removing them through endocytosis. This dynamic regulation is famously seen in the action of insulin, which triggers the insertion of glucose transporters into cell membranes to lower blood sugar levels after a meal.
Clinical Significance and Disease States
The medical implications of facilitated diffusion are vast and touch upon numerous specialties. Many genetic diseases are the direct result of mutations in the genes that code for transport proteins. For instance, cystic fibrosis is caused by a defect in a chloride ion channel (CFTR). This failure leads to thick, sticky mucus in the lungs and digestive tract because the proper balance of ions and water cannot be maintained across cell membranes. Understanding the structure and function of these channels is essential for developing new gene therapies and medications to correct these biological malfunctions.
In the management of diabetes mellitus, the principles of facilitated diffusion are central to clinical practice. Insulin acts as a signaling molecule that prompts muscle and fat cells to increase their rate of glucose uptake. When this signaling fails or the cells become resistant, glucose cannot efficiently enter the cells via facilitated diffusion, leading to hyperglycemia. Pharmacological research continues to focus on ways to sensitize these transport pathways or develop alternative ways to stimulate the recruitment of carrier proteins to the cell surface. These insights allow for better patient outcomes and the prevention of the long-term systemic damage associated with metabolic disorders.
Summary of Passive Transport Efficiency
In conclusion, facilitated diffusion represents an elegant solution to the problem of membrane impermeability. By utilizing the existing concentration gradient of the environment, cells can transport large volumes of polar and charged substances with high specificity and zero energy cost. This efficiency allows complex organisms to sustain life without the massive caloric burden that would be required if every molecule had to be actively pumped into the cell. From the simple movement of water via aquaporins to the complex regulation of neurotransmitters in the brain, facilitated diffusion is a silent, constant force that ensures the biochemical harmony of the human body. As medical science advances, our ability to manipulate these pathways will continue to lead to more effective treatments for vascular, neurological, and endocrine diseases.

