Apicomplexan protists comprise a diverse phylum of obligate intracellular parasites that cause significant human and animal diseases, most notably malaria, toxoplasmosis, and cryptosporidiosis. These organisms are unified by the presence of a specialized apical complex at one end of the cell, which enables host cell invasion, gliding motility, and establishment of a protected intracellular niche. Understanding the detailed ultrastructure of apicomplexan protists, as illustrated in the diagram, is fundamental to developing targeted therapies and vaccines against these globally important pathogens that continue to challenge public health systems worldwide.
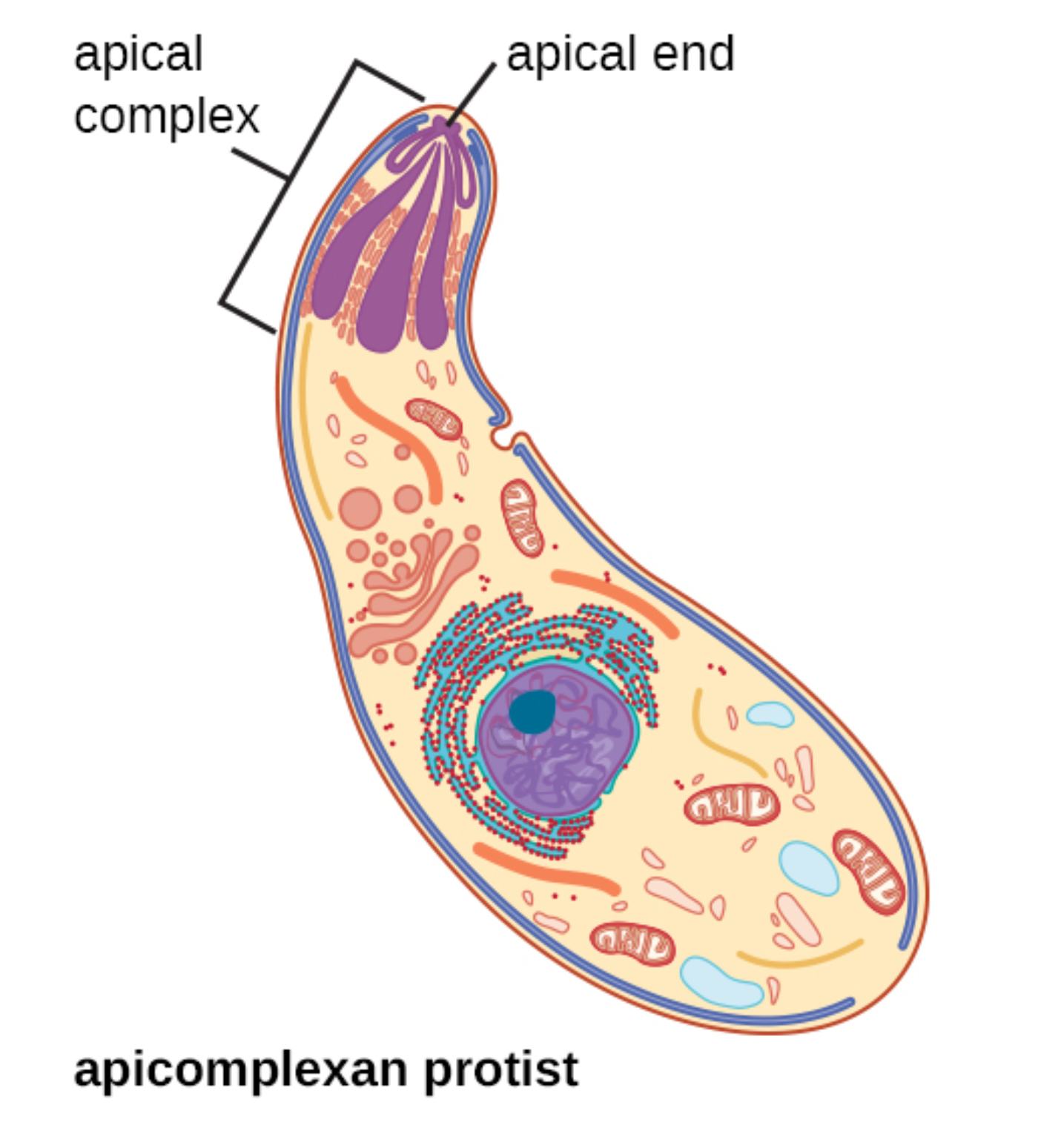
Apical complex is the defining feature of the phylum Apicomplexa, consisting of specialized secretory organelles and cytoskeletal elements concentrated at the anterior pole of the parasite. It includes rhoptries, micronemes, and associated structures that sequentially release proteins essential for host cell attachment, penetration, and formation of the parasitophorous vacuole. This complex allows apicomplexans to actively invade host cells without relying on host phagocytosis mechanisms.
Apical end refers to the tapered anterior region of the apicomplexan protist where the apical complex is localized. This polarized end directs the secretion of invasion-related molecules and orients the parasite during gliding motility and host cell entry, ensuring efficient invasion of target cells such as hepatocytes or erythrocytes in the case of Plasmodium species.
Apicomplexan protist describes the characteristic banana- or crescent-shaped cell depicted in the illustration, representing motile stages like merozoites, tachyzoites, or sporozoites. These cells possess a pellicle composed of the plasma membrane and inner membrane complex supported by subpellicular microtubules, which maintain shape and facilitate gliding motility powered by an actin-myosin motor system unique to the group.
Key Ultrastructural Features of Apicomplexan Protists
The diagram highlights the polarized organization typical of apicomplexan zoites. At the apical end, the complex includes club-shaped rhoptries and smaller micronemes that store adhesins and perforin-like proteins. The large nucleus with a prominent nucleolus occupies a central position, while mitochondria and other organelles support the high metabolic demands of invasion and intracellular replication. Subpellicular microtubules extend posteriorly from the apical polar ring, providing rigidity and enabling the characteristic gliding movement observed in live imaging studies.
Function of the Apical Complex in Parasite Invasion
The apical complex orchestrates a sophisticated, sequential secretion process during host cell invasion. Micronemes discharge first, releasing adhesins that mediate initial attachment and gliding motility. Rhoptries then secrete proteins that form the moving junction, remodel the host membrane, and establish the parasitophorous vacuole in which the parasite resides and replicates safely. This machinery is conserved across apicomplexans but shows species-specific adaptations, making it an attractive target for broad-spectrum interventions.
Major Diseases Caused by Apicomplexan Parasites
Apicomplexans are responsible for several high-burden infectious diseases. Plasmodium species cause malaria, with Plasmodium falciparum being the most lethal, leading to hundreds of thousands of deaths annually, primarily in sub-Saharan Africa. Toxoplasma gondii infects approximately one-third of the global population and can cause severe congenital defects or encephalitis in immunocompromised individuals. Cryptosporidium species cause debilitating diarrheal disease, particularly in young children and people living with HIV.
- Malaria transmission relies on Anopheles mosquitoes injecting sporozoites that use the apical complex to invade liver cells.
- Toxoplasma tachyzoites actively penetrate nearly any nucleated cell using apical complex-mediated invasion.
- Cryptosporidium oocysts are highly resistant to chlorination, complicating waterborne transmission control.
These parasites often manipulate host cell functions, including immune evasion and nutrient acquisition, to support their intracellular lifestyle.
Gliding Motility and Cytoskeletal Adaptations
Unlike many protists that use flagella or cilia, apicomplexans employ gliding motility driven by the actomyosin motor located beneath the pellicle. Adhesins secreted from the apical end link the parasite cytoskeleton to the substrate or host cell surface and are translocated rearward, propelling the organism forward. Subpellicular microtubules and the inner membrane complex provide the structural framework for this unusual form of movement, which is essential for tissue traversal and host cell invasion.
Therapeutic Targets Within Apicomplexan Structure
The unique features of apicomplexan protists, absent in human cells, offer selective drug targets. The apicoplast, a non-photosynthetic plastid-derived organelle, supports essential metabolic pathways inhibited by antibiotics like doxycycline. Components of the apical complex and invasion machinery are under investigation for vaccine development, including antigens from rhoptries and micronemes. Artemisinin-based therapies target blood-stage parasites, while new compounds aim to disrupt gliding or rhoptry secretion.
Diagnostic Approaches for Apicomplexan Infections
Diagnosis typically involves microscopic examination of blood smears for malaria, serological tests for toxoplasmosis, or acid-fast staining and PCR for cryptosporidiosis. Electron microscopy, as referenced in research on sporozoite ultrastructure, reveals fine details of the apical complex useful in laboratory studies. Molecular diagnostics increasingly allow species-specific identification and detection of drug resistance markers.
Prevention and Control Strategies
Effective control requires integrated approaches combining vector management, chemotherapy, and vaccines. Insecticide-treated nets and indoor residual spraying reduce malaria transmission. Hygiene measures limit fecal-oral spread of Cryptosporidium and Toxoplasma. The RTS,S and R21 malaria vaccines target sporozoite surface proteins to block liver invasion, demonstrating the translational value of understanding apical complex biology.
Research Advances and Future Perspectives
Genetic tractability of model organisms like Toxoplasma gondii and Plasmodium has accelerated discovery of apical complex components and their functions. Cryo-electron tomography and live imaging continue to refine models of invasion mechanics. Challenges such as artemisinin partial resistance and the need for transmission-blocking interventions drive ongoing research into novel targets within the conserved apicomplexan machinery.
Conclusion: The Central Role of Apicomplexan Ultrastructure in Parasitology
The specialized structure of the apicomplexan protist, centered on the apical complex and apical end, underpins the success of these parasites as intracellular pathogens. From enabling efficient host cell invasion to supporting complex life cycles, these features explain their medical significance and offer promising avenues for intervention. Continued study of apicomplexan biology remains essential for reducing the global burden of malaria, toxoplasmosis, and related diseases through improved diagnostics, therapeutics, and preventive measures.

