The life cycle of Dictyostelium discoideum beautifully illustrates the transition from solitary unicellular existence to cooperative multicellular development, offering profound lessons for cell biology, developmental processes, and human disease research. As a soil-dwelling cellular slime mold in the Amoebozoa supergroup, this organism serves as a recognized non-mammalian model by the National Institutes of Health for studying conserved eukaryotic mechanisms relevant to immunity, chemotaxis, neurodegeneration, and host-pathogen interactions. Its haploid genome, genetic tractability, and rapid 24-hour developmental program under starvation make it an invaluable tool for uncovering principles that apply to more complex systems, including human cells.
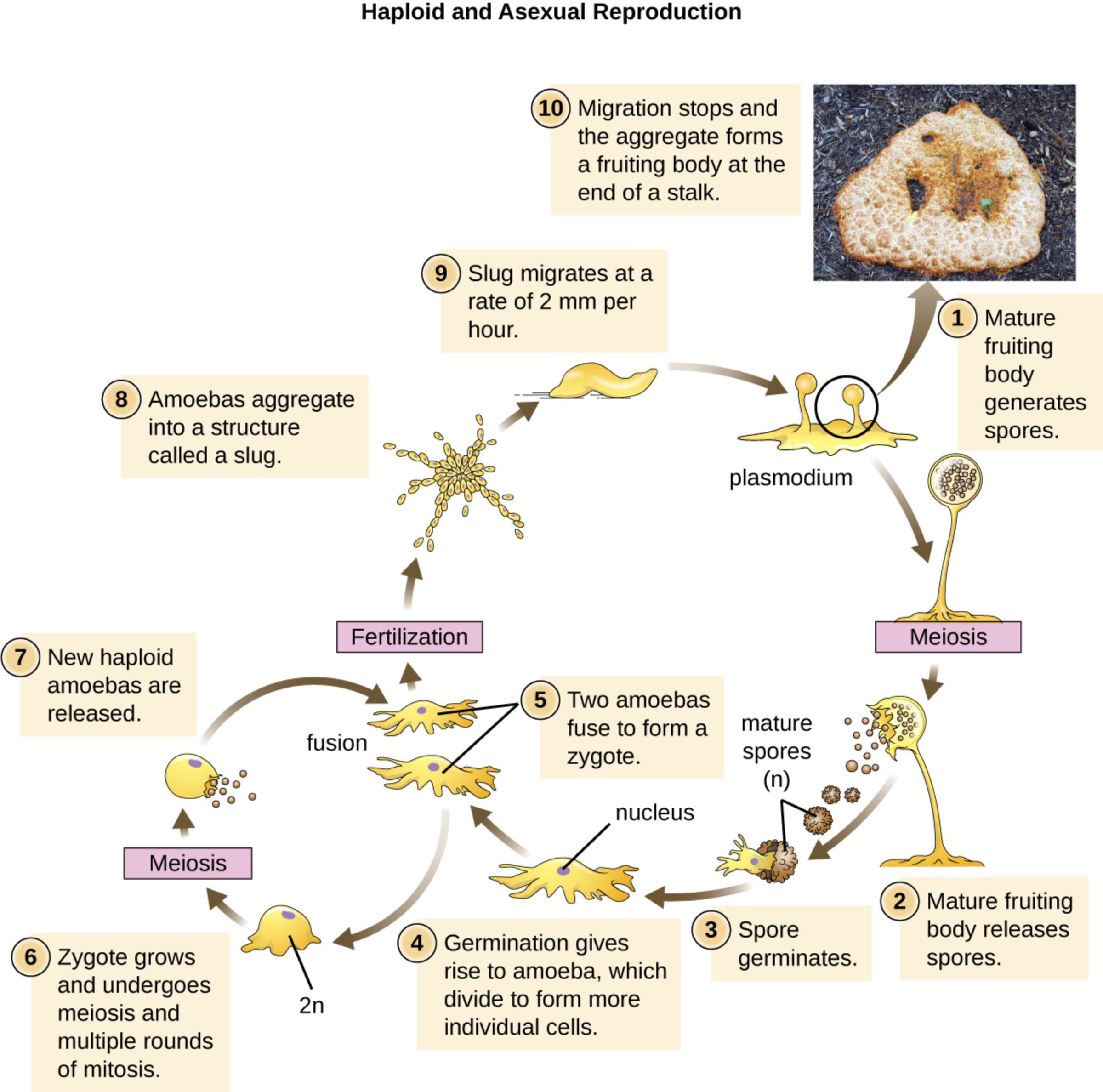
Mature fruiting body is the final multicellular structure formed at the end of development, consisting of a slender stalk supporting a sorus containing thousands of dormant spores. It represents the culmination of cellular differentiation and altruism, where some cells sacrifice themselves to elevate spores for better dispersal in the environment.
Plasmodium in this context refers to the early multicellular aggregate or slug-like structure formed during aggregation, though the term is sometimes used for the multinucleate mass in related slime molds. In Dictyostelium, it highlights the coordinated behavior of thousands of individual amoebae functioning as a single entity during migration.
Slug is the migratory multicellular stage that forms after aggregation, moving at approximately 2 mm per hour in response to environmental cues like light and humidity. This phase demonstrates collective cell migration, polarity, and differentiation into prestalk and prespore cells, providing a simple model for tissue morphogenesis and cell fate decisions.
Amoebas aggregate describes the critical step where solitary amoebae stream together in response to cyclic AMP signals to form the slug. This chemotactic aggregation is one of the best-studied examples of intercellular signaling and serves as a paradigm for understanding directed cell movement in development and immune responses.
New haploid amoebas are released from germinated spores, restarting the vegetative growth phase. These cells feed on bacteria via phagocytosis and divide mitotically until nutrients are depleted, initiating the developmental cycle again.
Zygote forms when two amoebae fuse during the sexual cycle, representing the diploid (2n) stage. The zygote undergoes meiosis and multiple rounds of mitosis to produce new haploid amoebae, though the asexual cycle predominates in laboratory studies.
Two amoebas fuse to form a zygote illustrates the rare sexual phase involving cell fusion and fertilization. This step introduces genetic recombination, contributing to diversity, while the primary laboratory focus remains on the asexual haploid cycle.
Germination gives rise to amoeba occurs when a spore breaks dormancy under favorable conditions, releasing a haploid amoeba that begins feeding and dividing. This transition links the resistant spore stage back to active vegetative growth.
Spore germinates refers to the process by which mature spores activate and emerge as new amoebae. Spores are highly resistant structures that ensure survival during unfavorable conditions.
Mature spores (n) are the haploid, dormant cells produced within the fruiting body sorus. They are released to colonize new areas, completing the asexual reproductive cycle.
Fertilization and Meiosis mark the sexual components of the life cycle, allowing genetic exchange. Meiosis restores the haploid state after zygote formation, while the dominant asexual pathway relies on mitosis for proliferation.
Overview of the Haploid and Asexual Life Cycle
The diagram emphasizes the predominant haploid asexual reproduction of Dictyostelium discoideum. In nutrient-rich conditions, individual amoebae grow and divide by mitosis. Starvation triggers the release of cyclic AMP pulses, leading to chemotactic aggregation into a mound that elongates into a migratory slug. The slug eventually forms a fruiting body, where cells differentiate: prestalk cells form the stalk and undergo programmed cell death, while prespore cells become viable spores. This altruistic division of labor provides insights into cell differentiation and cooperation.
Cellular Mechanisms Revealed by the Life Cycle
Each stage of the Dictyostelium discoideum life cycle highlights conserved eukaryotic processes. Chemotaxis during aggregation relies on G-protein-coupled receptors and PI3K signaling pathways similar to those in human neutrophils. Cytoskeletal dynamics drive pseudopod formation and cell motility, while cell adhesion molecules enable multicellular organization. These features make the organism ideal for dissecting fundamental cell biology applicable to wound healing, immune function, and cancer metastasis.
- Aggregation involves precise temporal and spatial signaling via extracellular cAMP.
- Slug migration integrates environmental sensing with collective movement.
- Terminal differentiation includes programmed cell death in stalk cells, analogous to apoptosis.
Such mechanisms are studied using live imaging, genetic mutants, and pharmacological interventions.
Dictyostelium discoideum as a Biomedical Model Organism
Recognized by the NIH as a key non-mammalian model, Dictyostelium discoideum bridges basic cell biology and human disease research. Its genome encodes homologs of many human genes linked to cancer, neurodegeneration, and immune disorders. The separation of growth and developmental phases allows independent analysis of proliferation versus differentiation, while the haploid nature simplifies gene knockout studies. Researchers use it to investigate host-pathogen interactions, as the amoebae naturally phagocytose bacteria in ways resembling macrophages.
Applications in Chemotaxis and Cell Motility Research
The aggregation and slug stages provide a powerful system for studying chemotaxis. Cells respond to cAMP gradients through surface receptors, activating intracellular cascades that regulate actin polymerization and directed movement. These pathways are conserved with human immune cell chemotaxis, offering insights into inflammation, autoimmune diseases, and tumor cell invasion. Quantitative analysis of mutant strains reveals the roles of specific signaling components.
Insights into Neurodegenerative Diseases
Dictyostelium discoideum has emerged as a valuable model for neurological disorders including Alzheimer’s, Parkinson’s, and Huntington’s diseases. It expresses homologs of proteins involved in protein aggregation, autophagy, and mitochondrial function. Researchers introduce human disease-associated proteins like alpha-synuclein or Tau to study their effects on cell behavior and development. The organism’s rapid life cycle enables high-throughput screening of potential therapeutic compounds affecting conserved pathways.
Host-Pathogen Interactions and Innate Immunity
As a professional phagocyte, Dictyostelium discoideum models innate immune responses and bacterial pathogenesis. It is used to study intracellular survival of pathogens such as Legionella and Mycobacterium, revealing virulence factors and host defense mechanisms. Findings often translate to understanding human macrophage function and infectious diseases, including how pathogens evade killing within phagosomes.
Advantages for Laboratory Research and Drug Screening
The organism grows easily in axenic media or on bacterial lawns, with a fully sequenced genome available through dictyBase. Genetic tools support targeted knockouts, overexpression, and fluorescent tagging. The 24-hour developmental cycle allows rapid phenotypic screening, while the non-sentient nature supports ethical research. These attributes facilitate pharmacogenetic studies and identification of drug targets for human conditions.
Broader Implications for Developmental and Evolutionary Biology
The life cycle demonstrates the evolution of multicellularity through aggregation rather than cell division, raising questions about altruism, cheating, and kin selection. Comparative studies with other amoebozoa and opisthokonts clarify eukaryotic evolutionary relationships. The model also informs understanding of developmental toxicity and teratogenesis, serving as a non-animal alternative for screening compounds.
Future Directions in Dictyostelium Research
Advances in CRISPR editing, single-cell transcriptomics, and high-resolution imaging will deepen insights into decision-making during development. Integrating findings with human iPSC models will enhance translational value. As antibiotic resistance and neurodegenerative diseases increase, Dictyostelium discoideum will continue contributing to innovative solutions in biomedical science through its accessible and conserved biology.
Conclusion: A Simple Organism with Complex Lessons
The life cycle of Dictyostelium discoideum, from solitary amoebae to cooperative fruiting bodies, encapsulates essential principles of eukaryotic cell behavior and multicellular organization. As a versatile biomedical model, it advances knowledge in chemotaxis, immunity, development, and disease mechanisms while offering practical advantages for research and screening. Continued exploration of this remarkable organism promises further breakthroughs benefiting human health and our understanding of life’s complexity.

