Trypanosoma brucei is a flagellated protozoan parasite responsible for human African trypanosomiasis, commonly known as sleeping sickness, a neglected tropical disease transmitted by the bite of infected tsetse flies in sub-Saharan Africa. The complex life cycle of this parasite alternates between the tsetse fly vector and mammalian hosts, including humans, involving multiple morphological transformations and multiplication by binary fission. The illustrated diagram clearly depicts the key stages in both the tsetse fly and human, providing essential understanding for medical professionals managing this potentially fatal infection that progresses from an initial hemolymphatic stage to a severe neurological phase if untreated.
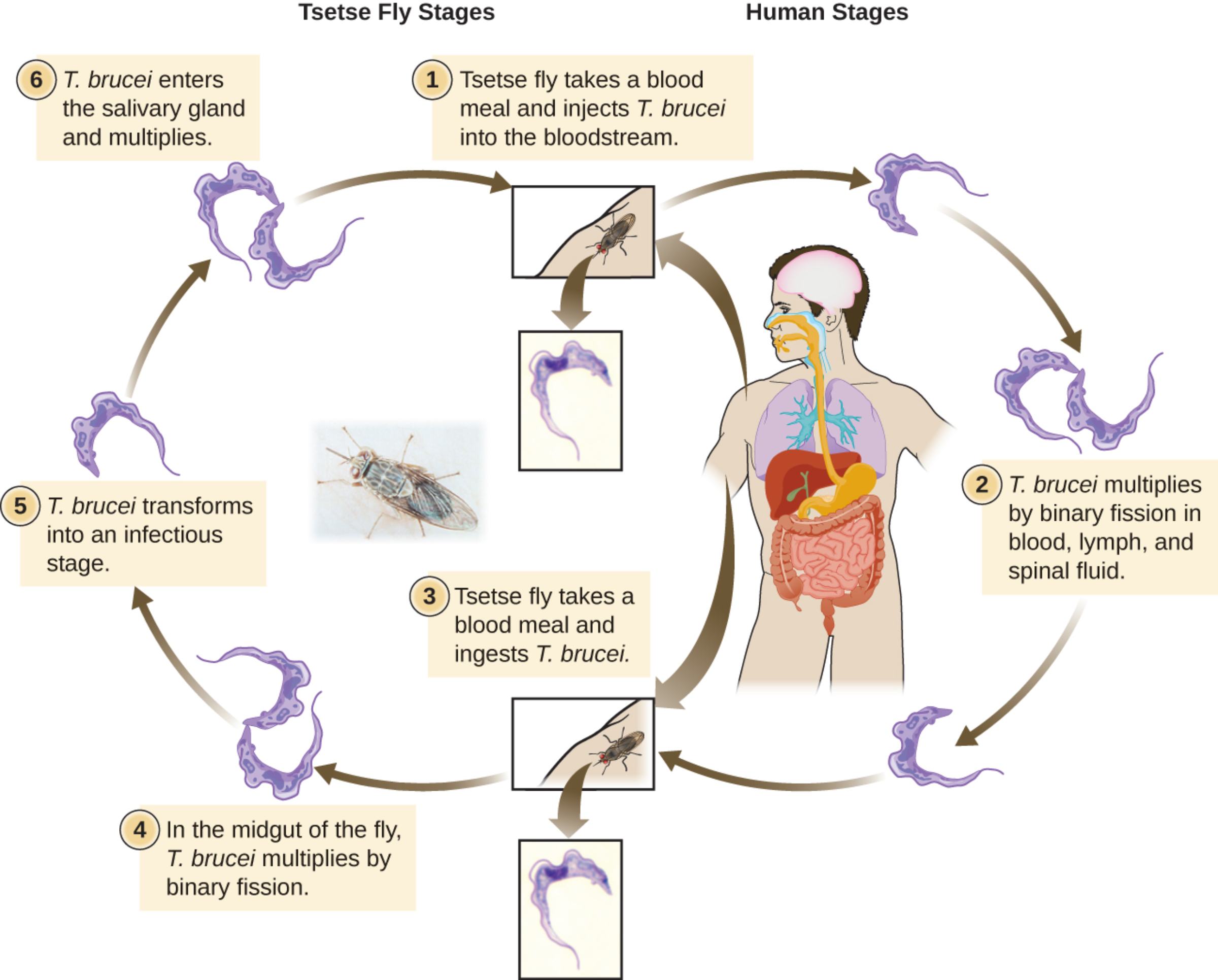
Tsetse fly takes a blood meal and injects T. brucei into the bloodstream represents the transmission step where an infected tsetse fly (Glossina species) bites a human or animal host. During the blood meal, metacyclic trypomastigotes are injected into the skin, entering the lymphatic system and bloodstream to initiate infection in the human host.
T. brucei multiplies by binary fission in blood, lymph, and spinal fluid describes the proliferative phase in the human host. Bloodstream trypomastigotes multiply extracellularly by binary fission, spreading through blood, lymph, and eventually crossing into the central nervous system, causing progressive symptoms.
Tsetse fly takes a blood meal and ingests T. brucei is the acquisition stage in the vector. When the tsetse fly feeds on an infected host, it ingests bloodstream trypomastigotes along with the blood meal, allowing the parasite to continue its development inside the fly.
In the midgut of the fly, T. brucei multiplies by binary fission refers to the procyclic stage in the tsetse fly midgut. Ingested trypomastigotes transform into procyclic forms that multiply by binary fission in the midgut before migrating to other parts of the fly.
T. brucei transforms into an infectious stage indicates the development of epimastigotes and subsequent differentiation into metacyclic trypomastigotes in the salivary glands, the form capable of infecting a new mammalian host upon the next bite.
T. brucei enters the salivary gland and multiplies describes the final maturation in the tsetse fly. Epimastigotes attach to the salivary gland epithelium, multiply, and transform into infectious metacyclic trypomastigotes ready for transmission during the next blood meal.
Life Cycle Overview of Trypanosoma brucei
The diagram illustrates the cyclical development of Trypanosoma brucei between the tsetse fly vector and human host. In the human, injected metacyclic trypomastigotes transform into bloodstream forms that multiply by binary fission, spreading systemically. When a tsetse fly takes a blood meal from an infected host, it ingests these forms. Inside the fly midgut, parasites transform into procyclic trypomastigotes and multiply. They then migrate, undergo further differentiation into epimastigotes in the salivary glands, and finally become metacyclic forms capable of infecting a new host. The entire cycle in the fly takes approximately three weeks.
Human Stages and Clinical Progression
In humans, the infection progresses in two main stages. The first hemolymphatic stage involves parasite multiplication in blood and lymph, causing intermittent fever, headache, joint pain, and swollen lymph nodes. If untreated, parasites cross the blood-brain barrier, entering the second meningoencephalitic stage characterized by sleep disturbances, confusion, behavioral changes, and neurological deterioration leading to coma and death. The diagram highlights multiplication in blood, lymph, and spinal fluid during this progression.
Tsetse Fly Stages and Vector Biology
The tsetse fly plays a critical role as the biological vector. After ingesting trypanosomes during a blood meal, parasites undergo transformation and multiplication in the midgut, followed by migration to the salivary glands where they become infectious metacyclic forms. Only a small percentage of flies become infected, and the cycle requires specific environmental conditions. Control of tsetse flies remains a cornerstone of disease prevention in endemic areas.
- Procyclic forms develop in the midgut after ingestion.
- Epimastigotes attach and multiply in salivary glands.
- Metacyclic trypomastigotes are the infective form injected into humans.
Understanding these stages is essential for interrupting transmission through vector control.
Medical Importance and Global Burden
Human African trypanosomiasis, caused primarily by Trypanosoma brucei gambiense (chronic form) and T. b. rhodesiense (acute form), remains a significant public health challenge in rural sub-Saharan Africa. The disease has two subspecies with distinct epidemiology and clinical courses. Early diagnosis and treatment are critical, as the neurological stage is difficult to manage and often fatal without intervention. Control efforts have reduced cases dramatically, but sustained surveillance is necessary to prevent resurgence.
Diagnosis, Treatment, and Prevention
Diagnosis involves serological screening followed by microscopic confirmation of parasites in blood, lymph node aspirate, or cerebrospinal fluid. Staging requires lumbar puncture to assess central nervous system involvement. Treatment depends on the stage and subspecies, with drugs such as pentamidine or fexinidazole for early gambiense disease and more toxic options like melarsoprol for late-stage disease. Prevention relies on tsetse fly control, personal protection, and active case finding in endemic communities.
Conclusion: The Complex Biology of Trypanosoma brucei
The life cycle diagram of Trypanosoma brucei elegantly captures the intricate interplay between the parasite, tsetse fly vector, and human host. From injection into the bloodstream to multiplication in tissues and return to the fly, each labeled stage underscores the parasite’s remarkable adaptations for survival and transmission. Continued research into these stages supports improved diagnostics, safer treatments, and effective control strategies aimed at eliminating this devastating disease from affected regions.

