The journey of human embryonic development unveils the remarkable beginnings of the cardiovascular system, with the heart starting to take shape by day 20 post-fertilization. This image highlights the critical endocardial tubes and the initiation of blood flow, offering a glimpse into the early circulatory dynamics that sustain embryonic growth and lay the foundation for a fully functional heart.
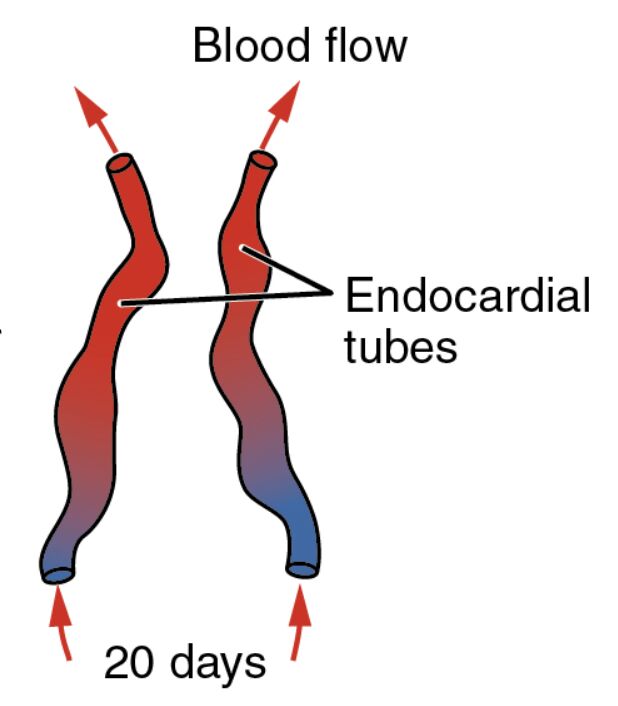
Blood flow Blood flow indicates the movement of primitive blood through the developing cardiovascular system, driven by the early contractions of the forming heart. This process is essential for delivering nutrients and oxygen to the growing embryo, marking the onset of circulation before the heart fully matures.
Endocardial tubes Endocardial tubes are the paired structures that form the initial heart tube, derived from the splanchnic mesoderm in the cardiogenic area. These tubes will eventually fuse and begin rhythmic contractions, setting the stage for the heart’s four-chambered structure in later development.
20 days 20 days signifies the gestational age of the embryo, corresponding to Carnegie stage 9, where significant cardiovascular milestones occur. At this point, the embryo is approximately 2-3 mm long, with the heart tube starting to elongate and bend, a precursor to its complex looping process.
The Role of Early Cardiovascular Structures
This phase of embryonic development showcases the emergence of the heart’s foundational elements. The image captures a pivotal moment where simple tubes transform into a beating organ, critical for survival.
- Formation of Endocardial Tubes: These tubes arise from the fusion of endothelial-lined channels within the cardiogenic mesoderm. Their alignment and merging are guided by molecular signals like VEGF and FGF, ensuring proper heart tube assembly.
- Initiation of Blood Flow: The arrows depict the direction of primitive blood movement, facilitated by spontaneous contractions of the heart tube. This early circulation relies on the yolk sac for oxygenation until the placenta takes over.
- Embryonic Environment: The embryo resides in a fluid-filled amniotic cavity, protecting these delicate structures. Nutrient exchange occurs via diffusion and the vitelline circulation, supporting rapid cellular growth.
Anatomical Insights into the 20-Day Embryo
The illustration reveals the embryo’s elongated shape, with the endocardial tubes positioned centrally as the heart begins to form. Color gradients and arrows provide a clear visual of blood dynamics at this stage.
At 20 days, the embryo undergoes significant folding, bringing the cardiogenic region into the thoracic area. This morphogenetic event is crucial for aligning the heart with other developing systems.
- Structure of Endocardial Tubes: Each tube consists of an inner endothelial layer surrounded by myocardial cells, which will later thicken to form the heart walls. The fusion process eliminates the midline septum, creating a single heart tube.
- Blood Flow Dynamics: The sluggish flow reflects the immature nature of the vascular network, with primitive erythrocytes carrying limited oxygen. This flow pattern helps establish pressure gradients for further vessel development.
- Supporting Tissues: The surrounding splanchnic mesoderm provides structural support and contributes to the epicardium. This layer will eventually cover the heart, aiding in its protection and nourishment.
Developmental Milestones at 20 Days
Significant progress marks the embryo at this stage, with the cardiovascular system taking center stage. The heart’s early beating signals the transition from passive diffusion to active circulation.
The neural tube continues to close, while somites increase in number along the body axis. These developments occur alongside cardiovascular maturation, reflecting the coordinated nature of embryogenesis.
- Heart Tube Elongation: Following fusion, the heart tube lengthens and begins to loop, a process driven by differential growth rates. This looping establishes the future left-right asymmetry of the heart.
- Genetic Regulation: Genes such as NKX2-5 and TBX5 are actively expressed, directing cardiomyocyte differentiation. Alterations in these genes can lead to congenital heart defects like ventricular septal defects.
- Cellular Contributions: Cardiac neural crest cells migrate to the outflow tract, contributing to septum formation. This migration is tightly regulated to prevent structural anomalies.
- Nutritional Support: The yolk sac remains the primary source of nutrients, with vitelline veins channeling blood back to the heart tube. This temporary system bridges the gap until placental circulation develops.
Physiological Functions of Early Circulation
The physiology of the 20-day embryo revolves around establishing a rudimentary circulatory system. The heart’s initial contractions mark the beginning of a lifelong process of blood propulsion.
This early system operates under low pressure, relying on the yolk sac for oxygenation. The transition to fetal circulation will refine these mechanisms over the coming weeks.
- Contraction Mechanisms: The myocardial layer initiates peristaltic-like movements, driven by calcium signaling. These contractions are spontaneous and increase in coordination as development progresses.
- Oxygen Delivery: Primitive red blood cells, produced in blood islands, carry hemoglobin with limited oxygen-binding capacity. This compensates for the low oxygen tension in the embryonic environment.
- Vascular Development: Angiogenic sprouting extends the primitive vascular network, guided by growth factors. This process ensures the heart tube integrates with the emerging arterial and venous systems.
- Future Adaptations: Shunts like the foramen ovale will develop to bypass the lungs, adapting circulation to the intrauterine environment. Understanding these adaptations aids in diagnosing fetal conditions.
Clinical and Research Perspectives
Studying the 20-day embryo provides valuable insights for medical practice and research. Early cardiovascular development is a critical window for identifying potential congenital issues.
Advances in embryonic imaging enhance our ability to monitor these stages. Stem cell research further explores how to replicate these structures for therapeutic purposes.
- Congenital Anomalies: Defects in endocardial tube fusion can result in conditions like double outlet right ventricle. Early ultrasound can detect such anomalies, guiding prenatal care.
- Regenerative Medicine: Stem cells can be differentiated into cardiomyocytes, mimicking the endocardial tube stage. This approach holds potential for repairing damaged hearts.
- Research Models: Zebrafish and chick embryos offer comparative insights due to similar cardiovascular development. These models help validate human findings.
- Therapeutic Targets: Modulating VEGF pathways may enhance vascular growth in congenital defects. Gene therapy targeting cardiac genes is an emerging field.
In conclusion, this image of the 20-day embryo highlights the intricate beginnings of the human heart, from the fusion of endocardial tubes to the onset of blood flow. These early structures are a testament to the precision of developmental biology, offering a foundation for both understanding health and advancing medical interventions.

