Apicomplexan protists, particularly members of the genus Plasmodium, are among the most medically important eukaryotic parasites, responsible for malaria that affects hundreds of millions of people annually. The sporozoite stage represents the infectious form injected by female Anopheles mosquitoes into the human bloodstream, initiating the complex life cycle that leads to severe disease. Detailed understanding of sporozoite ultrastructure, especially the specialized apical complex, is crucial for developing effective vaccines, antimalarial drugs, and control strategies against this devastating global health threat.
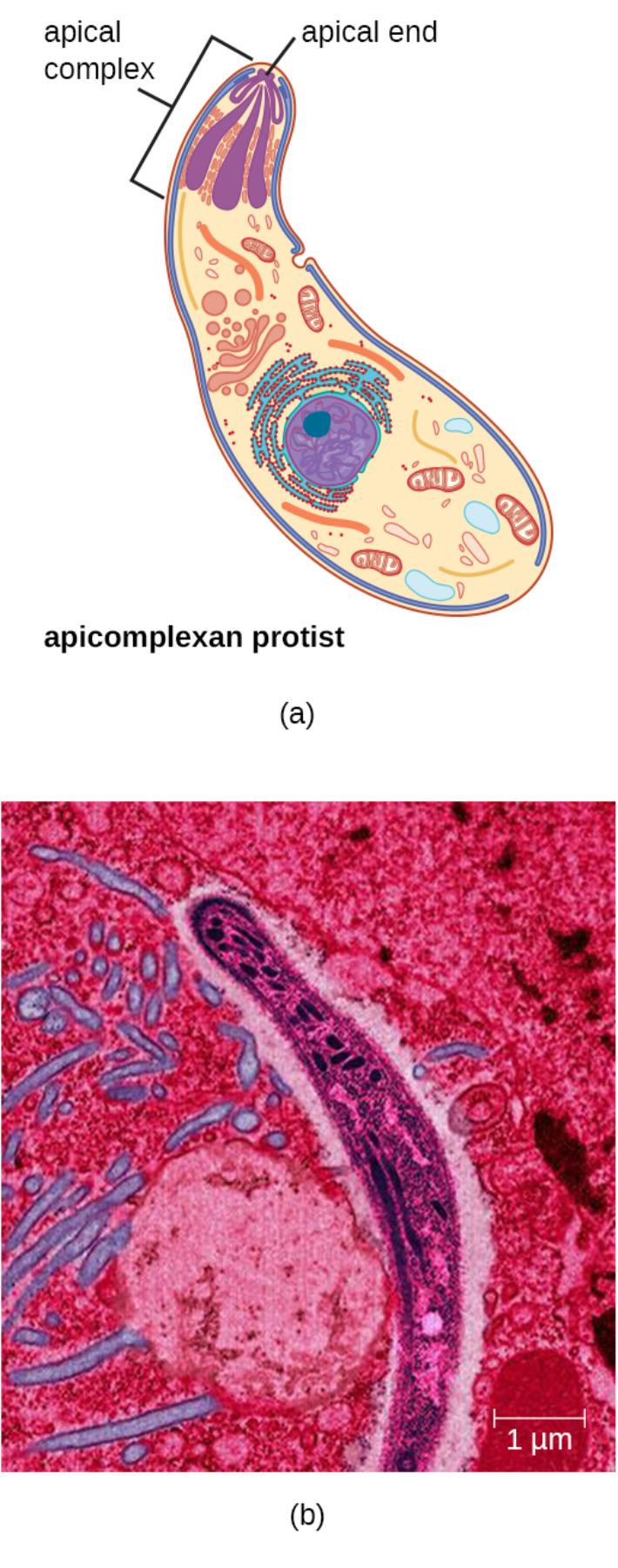
Apical complex is a unique set of organelles located at the anterior end of apicomplexan parasites. It includes structures such as rhoptries, micronemes, and a conoid that enable the parasite to invade host cells by secreting proteins that modify the host membrane and facilitate entry. This complex is essential for the sporozoite’s ability to penetrate liver cells and begin the exoerythrocytic phase of infection.
Apical end refers to the tapered anterior portion of the sporozoite where the apical complex is concentrated. This specialized pole directs the secretion of invasion-related proteins and orients the parasite during host cell attachment and penetration, playing a critical role in the initial stages of infection.
Apicomplexan protist describes the elongated, banana-shaped cell shown in the diagram, characteristic of Plasmodium sporozoites. These motile stages possess a pellicle consisting of inner membrane complex and microtubules that provide structural support and enable gliding motility, a form of movement unique to apicomplexans that does not rely on flagella or cilia.
Ultrastructure of the Plasmodium Sporozoite
The transmission electron micrograph and schematic illustration reveal key internal features of the sporozoite. The large central nucleus contains densely packed chromatin, while numerous mitochondria and other organelles support the high energy demands of invasion and migration. Rhoptries appear as elongated secretory organelles at the apical end, and micronemes store adhesins necessary for host cell recognition. The surface pellicle and subpellicular microtubules maintain the characteristic shape and facilitate gliding motility powered by an actin-myosin motor system.
The Role of the Apical Complex in Host Cell Invasion
The apical complex is the defining feature of the phylum Apicomplexa and is vital for the pathogenicity of Plasmodium species. Upon contact with a host cell, micronemes discharge adhesins that mediate attachment, followed by rhoptry secretion that forms the moving junction and modifies the parasitophorous vacuole membrane. This sophisticated invasion machinery allows the sporozoite to enter hepatocytes efficiently, where it develops into thousands of merozoites. Understanding these molecular events has driven research into invasion-blocking vaccines and drugs that target apical complex components.
Malaria Life Cycle and the Sporozoite Stage
In the malaria life cycle, sporozoites are produced in the mosquito midgut after sexual reproduction and migrate to the salivary glands. When the mosquito takes a blood meal, sporozoites are injected into the human skin and quickly enter the bloodstream to reach the liver. Inside hepatocytes, they undergo schizogony to produce merozoites that then infect erythrocytes, initiating the symptomatic blood stage. The sporozoite stage is a primary target for pre-erythrocytic vaccines because blocking invasion at this point prevents all subsequent pathology.
- Sporozoites travel rapidly through the skin and blood to reach the liver within minutes.
- They traverse several hepatocytes before settling in one for replication.
- The circumsporozoite protein on the surface is a major antigen in current malaria vaccines.
Disrupting sporozoite motility or invasion could dramatically reduce malaria transmission.
Diagnostic and Microscopic Identification
Sporozoites are rarely seen in routine clinical samples because they reside briefly in the blood before entering the liver. However, the electron micrograph in the image highlights ultrastructural details used in research laboratories to study parasite biology. In diagnostic settings, malaria is typically identified by observing ring forms, trophozoites, or gametocytes in blood smears rather than sporozoites. Advanced imaging and molecular techniques now allow detailed visualization of apical complex components during invasion studies.
Therapeutic and Vaccine Development Targeting Apicomplexans
The unique apical complex and gliding motility of Plasmodium sporozoites offer selective targets for intervention. Current research focuses on vaccines like RTS,S and R21 that elicit antibodies against the circumsporozoite protein to neutralize sporozoites before liver invasion. Novel drugs aim to inhibit actin-myosin motors or rhoptry secretion. Because the apical complex is conserved across apicomplexans, including Toxoplasma and Cryptosporidium, findings from Plasmodium research often apply to other clinically relevant parasites.
Global Health Impact of Apicomplexan Parasites
Malaria caused by Plasmodium falciparum and other species remains a leading cause of mortality in tropical and subtropical regions, particularly among children under five. The sporozoite stage is critical in transmission dynamics, making mosquito control and pre-erythrocytic interventions essential components of elimination strategies. Climate change and drug resistance continue to challenge control efforts, underscoring the need for deeper understanding of sporozoite biology.
Research Techniques and Future Directions
Modern techniques including cryo-electron tomography, live-cell imaging, and genetic manipulation of parasites have greatly advanced knowledge of sporozoite structure and function. Researchers are exploring how sporozoites navigate the skin and evade immune detection during their journey to the liver. Future therapies may combine sporozoite-targeting vaccines with blood-stage drugs and transmission-blocking interventions to achieve synergistic effects against malaria.
Conclusion: The Strategic Importance of Sporozoite Biology
The detailed structure of the apicomplexan protist Plasmodium sporozoite, particularly its apical complex, reveals the sophisticated adaptations that enable this parasite to initiate human infection. Continued research into these mechanisms is essential for developing next-generation tools to combat malaria and related apicomplexan diseases. By targeting the vulnerable sporozoite stage, the global health community moves closer to effective control and eventual elimination of one of humanity’s oldest and most persistent pathogens.

