The Plasmodium sporozoite represents the infective stage of the malaria parasite, transmitted by Anopheles mosquitoes directly into the human bloodstream or skin during a blood meal. This highly specialized, elongated cell must rapidly navigate host tissues, evade immune detection, and invade hepatocytes to initiate the asymptomatic liver stage of infection. Detailed ultrastructural studies, including transmission electron microscopy, reveal the sophisticated machinery that enables sporozoite motility and invasion, offering critical insights for malaria vaccine development and novel therapeutic strategies aimed at interrupting transmission at its earliest point.
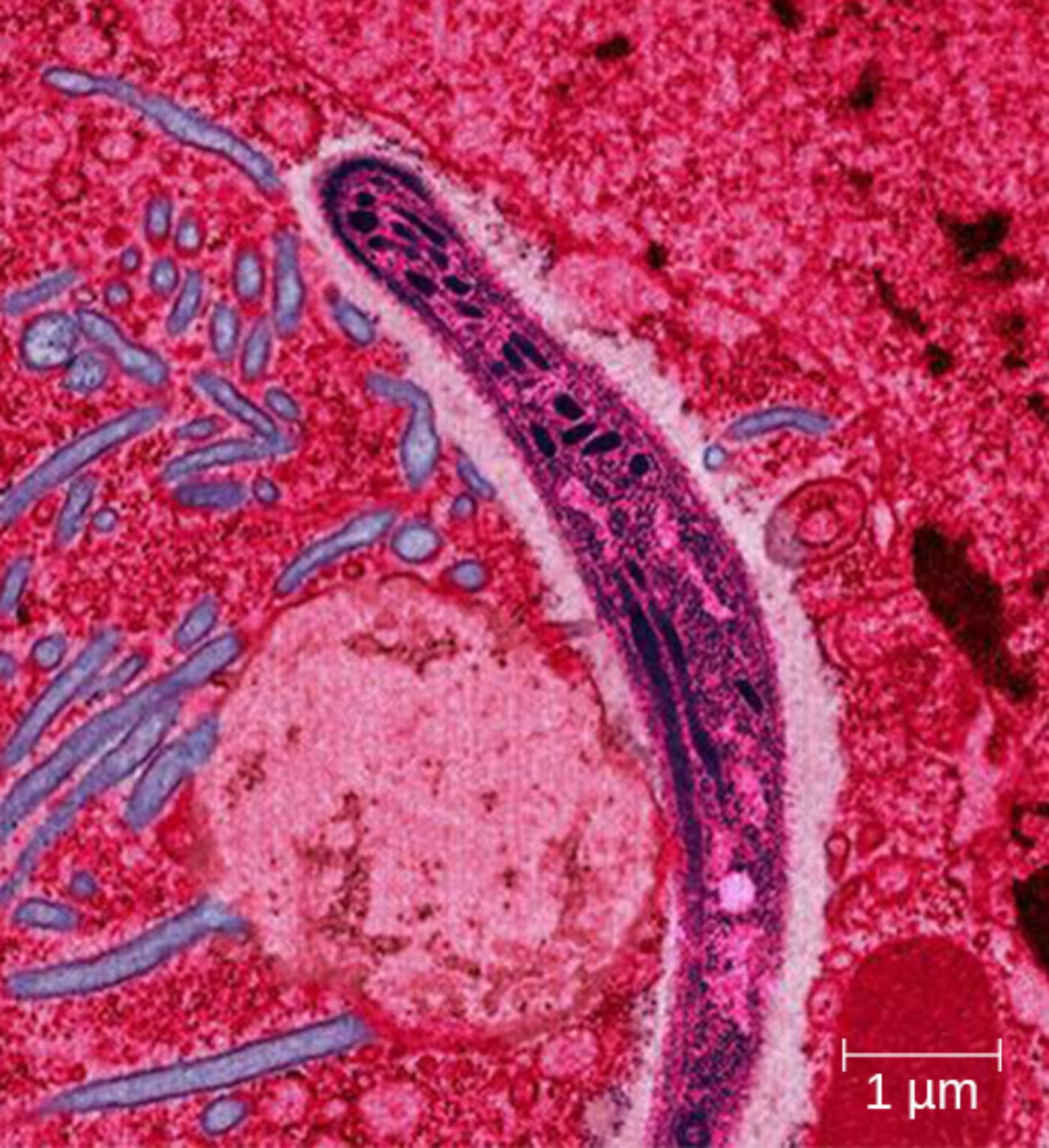
Plasmodium sporozoite is the slender, elongated infectious form of the malaria parasite visible in the electron micrograph. Measuring approximately 10-15 micrometers in length, it possesses a characteristic curved or banana-like shape supported by a pellicle and subpellicular microtubules that maintain structural integrity during rapid gliding motility through host tissues.
Apical complex is the specialized anterior machinery characteristic of apicomplexan parasites, though partially sectioned in this view. It includes rhoptries and micronemes that secrete proteins facilitating host cell recognition, attachment, and penetration, enabling the sporozoite to traverse skin and enter the liver efficiently.
1 μm scale bar provides essential context for the microscopic scale of the image. The bar indicates that the sporozoite cross-section and surrounding cellular structures are visualized at high resolution, highlighting fine ultrastructural details such as membranes and organelles within the parasite and host tissue environment.
Ultrastructure of the Plasmodium Sporozoite
The transmission electron micrograph captures a longitudinal or oblique section through a Plasmodium sporozoite within host tissue, likely liver or skin. The parasite appears as a dark, elongated structure with a distinct pellicle consisting of the plasma membrane and inner membrane complex. Internal features include the nucleus, mitochondria, and secretory organelles associated with the apical complex. Surrounding pink-stained host cells and extracellular matrix provide contrast, illustrating the intimate interaction between the sporozoite and host environment during invasion or traversal.
The Apical Complex in Sporozoite Invasion
The apical complex at the leading end of the sporozoite orchestrates a precise sequence of events for host cell entry. Micronemes release adhesins that support gliding motility, while rhoptries discharge proteins that remodel the host membrane to form the moving junction and parasitophorous vacuole. This machinery allows the sporozoite to actively penetrate hepatocytes without triggering immediate immune clearance, setting the stage for massive replication into merozoites.
Malaria Life Cycle and Sporozoite Role
Sporozoites are produced in the mosquito midgut following gamete fusion and migrate to the salivary glands. Upon injection into human skin, they must traverse dermal tissue, enter blood vessels or lymphatics, and reach the liver within minutes. Inside hepatocytes, each sporozoite develops into tens of thousands of merozoites over 5-16 days depending on the Plasmodium species. Blocking the sporozoite stage prevents all downstream blood-stage pathology and transmission.
- Sporozoites exhibit rapid gliding motility exceeding 1 μm per second.
- They can traverse multiple cells before productively invading a hepatocyte.
- The circumsporozoite protein coats the surface and is a primary vaccine target.
Understanding sporozoite biology is therefore central to pre-erythrocytic malaria control strategies.
Gliding Motility Mechanism
Sporozoites move by an actin-myosin motor system located beneath the pellicle. Adhesins secreted from the apical end link the motor to the substrate and are translocated rearward, propelling the parasite forward in a corkscrew-like motion. Subpellicular microtubules provide tracks and rigidity for this unique form of locomotion, which does not require flagella and enables efficient migration through extracellular matrix and cell barriers.
Challenges in Sporozoite-Targeted Interventions
While the sporozoite stage is vulnerable outside cells, its brief extracellular existence and rapid invasion pose challenges for immunity. Current vaccines like RTS,S and R21 target the circumsporozoite protein to elicit neutralizing antibodies that immobilize or opsonize sporozoites. However, efficacy remains moderate, driving research into multi-antigen approaches that also target apical complex components or invasion ligands.
Electron Microscopy in Malaria Research
Transmission electron microscopy, as shown in the image with its 1 μm scale bar, has been instrumental in elucidating sporozoite ultrastructure and invasion dynamics. Advanced techniques such as cryo-electron tomography now reveal three-dimensional organization of the apical rings, rhoptries, and microtubule arrangements at near-atomic resolution. These insights guide rational design of inhibitors that disrupt specific steps in motility or invasion.
Global Health Context of Plasmodium Infections
Malaria continues to cause significant morbidity and mortality, particularly in children and pregnant women in endemic regions. The sporozoite-initiated liver stage is clinically silent, allowing parasites to establish infection before symptoms appear. Integrated control combining vector management, chemoprevention, and vaccines aims to reduce transmission intensity and eventually achieve elimination in many areas.
Future Directions in Sporozoite Research
Ongoing studies focus on sporozoite traversal mechanisms, immune evasion strategies, and genetic diversity across Plasmodium species. Single-cell transcriptomics and advanced imaging promise to uncover new vulnerabilities in the apical complex and motility apparatus. Combining sporozoite-targeting vaccines with blood-stage and transmission-blocking interventions offers the best path toward sustainable malaria control.
Conclusion: Targeting the Sporozoite for Malaria Elimination
The intricate structure of the Plasmodium sporozoite, revealed through high-resolution electron microscopy, underscores its critical role as the gateway to malaria infection. From the apical complex enabling precise host cell invasion to the cytoskeletal adaptations supporting rapid motility, these features highlight both the parasite’s sophistication and potential weaknesses. Sustained research into sporozoite biology remains essential for developing more effective vaccines and therapies that can ultimately reduce the global burden of this ancient disease.

