Giardiasis is one of the most common waterborne parasitic infections worldwide, affecting millions of people annually across both developing and industrialized nations. At the heart of this condition are the microscopic Giardia lamblia, single-celled protozoans that have successfully adapted to survive in the harsh environment of the mammalian digestive tract. These parasites are notorious for their ability to cause significant gastrointestinal distress, often following the ingestion of contaminated water from seemingly pristine wilderness streams or poorly treated municipal supplies. Understanding the intricate biology and morphology of these organisms is the first step in recognizing the pathology they induce and implementing effective clinical interventions to restore patient health.
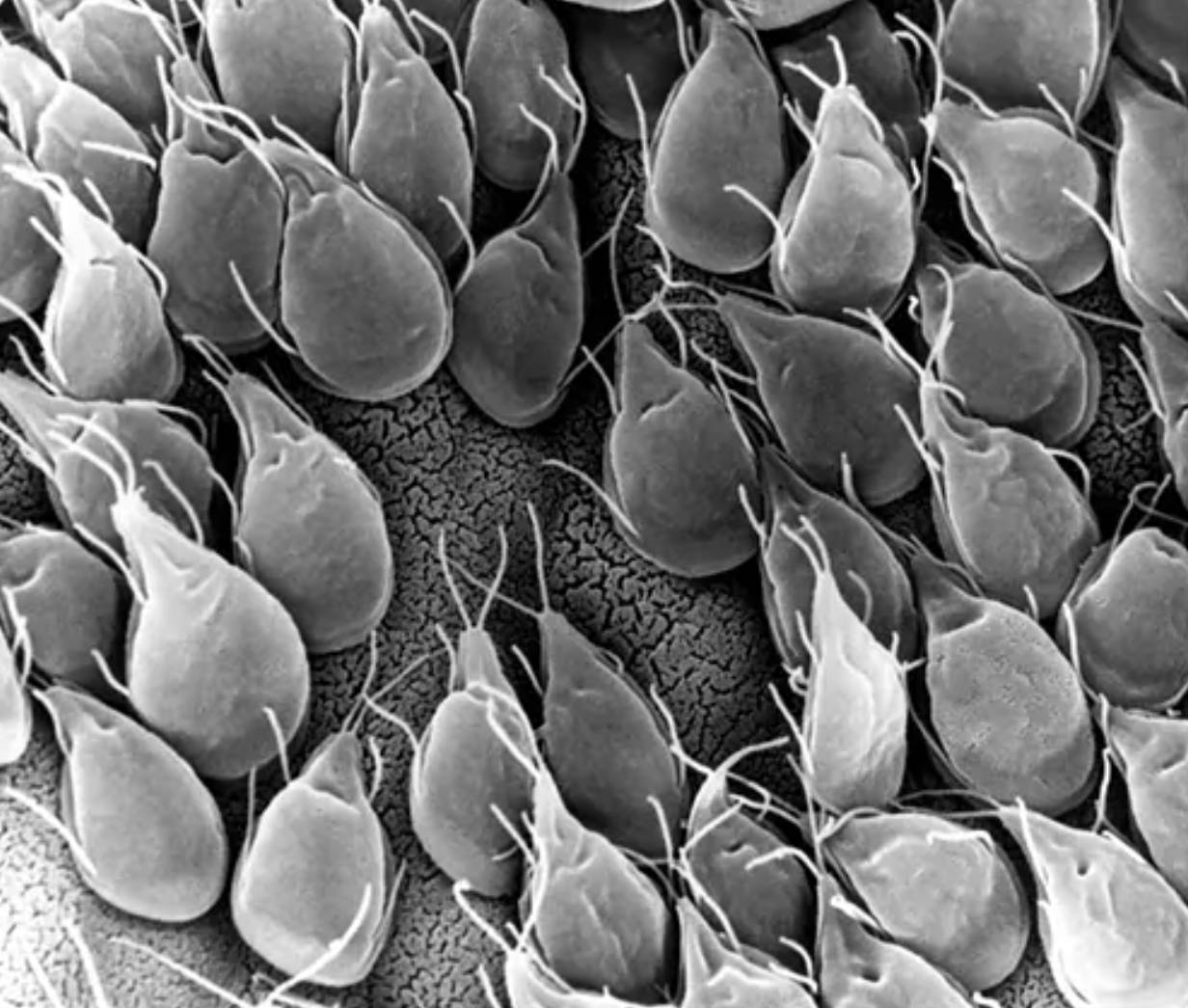
Trophozoite body: This represents the active, vegetative stage of the parasite which is pear-shaped and possesses a distinctive bilateral symmetry. In this scanning electron micrograph, the bodies are seen clustered together as they would appear when colonizing the mucosal surface of the small intestine.
Flagella: These are the long, thread-like appendages visible protruding from various points on the parasite cells, used primarily for locomotion. Each individual organism typically possesses four pairs of these flagella that allow it to swim through the intestinal lumen to find a suitable site for attachment.
Ventral sucking disk: Although the underside is not fully visible here due to attachment, the concave area on the anterior portion of the body acts as a powerful adhesive organ. This specialized structure allows the parasite to resist being washed away by the natural peristaltic movements of the gut.
Intestinal mucosa surface: The textured background upon which the parasites are resting indicates the epithelial lining of the host’s digestive system. The density of the parasites shown illustrates how they can physically carpet the lining, creating a barrier that interferes with the host’s ability to absorb nutrients effectively.
The Biological Profile of Giardia Parasites
The organism responsible for giardiasis, historically known as Giardia intestinalis or Giardia duodenalis, is a flagellated protozoan that lacks traditional mitochondria, instead possessing simplified organelles called mitosomes. This primitive cellular architecture suggests a long evolutionary history of adaptation to anaerobic or microaerophilic environments, such as the upper small intestine. When viewed under a light microscope using specific stains, the trophozoites often appear to have a “face,” created by the presence of two nuclei and the arrangement of the cytoskeleton, giving them a unique and easily identifiable appearance in clinical laboratory settings.
These parasites are highly specialized for life in the duodenum and jejunum. Their primary mechanism of survival involves the extraction of nutrients directly from the host’s predigested food. Unlike many other intestinal pathogens, they do not typically invade the tissue or enter the bloodstream; instead, they remain extracellular, attached firmly to the brush border of the intestinal villi. This attachment is not merely a physical presence; it triggers a cascade of physiological changes in the host, including the shortening of microvilli and the disruption of tight junctions between epithelial cells, which leads to the hallmark symptoms of the infection.
The Two-Stage Life Cycle and Transmission
To understand how this infection spreads, one must look at its dimorphic life cycle, which consists of the trophozoite and the cyst stages. The cysts are the infective, resistant form of the parasite, characterized by a thick, protective outer wall that allows them to survive for months in cold water and resist standard chlorine disinfection. Once a host ingests contaminated water or food containing these hardy structures, the acidic environment of the stomach triggers a process called excystation. During this transition, the cyst breaks open to release two trophozoites, which then migrate to the small intestine to begin the cycle of multiplication and attachment.
As the trophozoites move toward the colon, the changing environment—specifically the increase in bile salts and decrease in moisture—triggers encystation. The parasites transform back into cysts, which are then excreted in the feces, ready to infect a new host. This fecal-oral route is the primary method of transmission. It is particularly prevalent in settings with poor sanitation, in childcare facilities where diaper-changing practices may lead to hand-to-mouth transfer, and among outdoor enthusiasts who consume untreated water from lakes or rivers, a phenomenon often colloquially referred to as “beaver fever.”
Pathophysiology and the Development of Giardiasis
The clinical manifestation of the infection, known as giardiasis, is a result of both the physical presence of the parasites and the host’s inflammatory response. When thousands of trophozoites adhere to the intestinal wall, they create a physical barrier to absorption. More importantly, they cause the villi—the finger-like projections responsible for nutrient uptake—to become blunted or atrophied. This damage specifically affects the production of digestive enzymes like lactase, which is why many patients develop temporary lactose intolerance during and immediately after the infection.
The malabsorption of fats and carbohydrates leads to significant changes in stool composition. The unabsorbed nutrients draw water into the bowel through osmosis, resulting in diarrhea. Furthermore, the fermentation of unabsorbed carbohydrates by gut bacteria produces excess gas, leading to the characteristic bloating and foul-smelling flatulence associated with the disease. In chronic cases, this persistent malabsorption can lead to significant weight loss and nutritional deficiencies, particularly in children whose growth and development may be hindered by the lack of essential vitamins and calories.
Clinical Symptoms and Diagnostic Challenges
Symptoms usually appear one to three weeks after exposure and can vary significantly in severity. While some individuals remain asymptomatic carriers, most experience acute symptoms including watery, greasy, and foul-smelling stools, abdominal cramps, nausea, and dehydration. A unique feature of this infection is the lack of blood or mucus in the stool, which helps distinguish it from other types of infectious diarrhea like dysentery. The fatigue associated with the disease can be profound, often lasting longer than the gastrointestinal symptoms themselves.
Diagnosing the condition can be challenging because the shedding of cysts in the stool is often intermittent. A single stool sample may yield a false negative, which is why clinicians frequently request three separate samples collected over several days to increase the likelihood of detection. Modern diagnostic techniques have moved toward enzyme-linked immunosorbent assays (ELISA) and polymerase chain reaction (PCR) tests, which are much more sensitive and specific than traditional microscopic ova and parasite exams. These tests look for specific proteins or DNA sequences of the parasite, providing a faster and more reliable diagnosis for patients in acute distress.
Treatment Strategies and Pharmacotherapy
Once a diagnosis is confirmed, treatment focuses on eradicating the parasite and managing dehydration. The first-line medications are typically nitroimidazoles, such as metronidazole or tinidazole. These drugs work by interfering with the DNA synthesis of the anaerobic protozoan, effectively killing the trophozoites. While these medications are highly effective, they can cause side effects like a metallic taste in the mouth or nausea, and patients must strictly avoid alcohol during the course of treatment to prevent a disulfiram-like reaction.
In cases where the infection is resistant to standard therapy, or for specific patient populations like pregnant women, alternative medications such as nitazoxanide or paromomycin may be used. It is also essential to treat all members of a household if symptoms are present, as the parasite can easily cycle between family members. Following the completion of the medication, follow-up testing is rarely necessary unless symptoms persist, in which case the physician must evaluate for potential reinfection, treatment failure, or a secondary condition like post-infectious irritable bowel syndrome.
Prevention and Public Health Measures
Prevention is the most effective way to combat the spread of this parasite. For travelers and hikers, the gold standard is to boil water for at least one minute (or three minutes at high altitudes) to ensure that any cysts are destroyed. High-quality water filters with a pore size of 1 micron or smaller are also effective at physically removing the cysts. While chemical treatments like iodine or chlorine tablets can work, they require long contact times and may be less reliable in cloudy or very cold water.
On a broader scale, public health measures focus on improving water treatment facilities and maintaining strict hygiene standards in public settings. Handwashing with soap and water remains the single most effective personal hygiene practice, as alcohol-based hand sanitizers are not consistently effective against the hardy cyst stage of the parasite. By educating the public on the risks of untreated water and the importance of sanitation, the incidence of this pervasive parasitic infection can be significantly reduced, protecting the health of the community and the integrity of the human digestive system.

