Helicobacter pylori is a Gram-negative, microaerophilic bacterium that colonizes the gastric mucosa of approximately half the world’s population, playing a central role in the development of peptic ulcers, chronic gastritis, and gastric cancer. This spiral-shaped organism has evolved remarkable adaptations for surviving the harsh acidic environment of the stomach, including powerful motility and urease production. The provided scanning electron micrograph captures the distinctive ultrastructure of H. pylori, highlighting its helical body and polar flagella as it interacts with the textured surface of gastric epithelial cells or mucus, offering valuable visual insight into its morphology and pathogenic mechanisms.
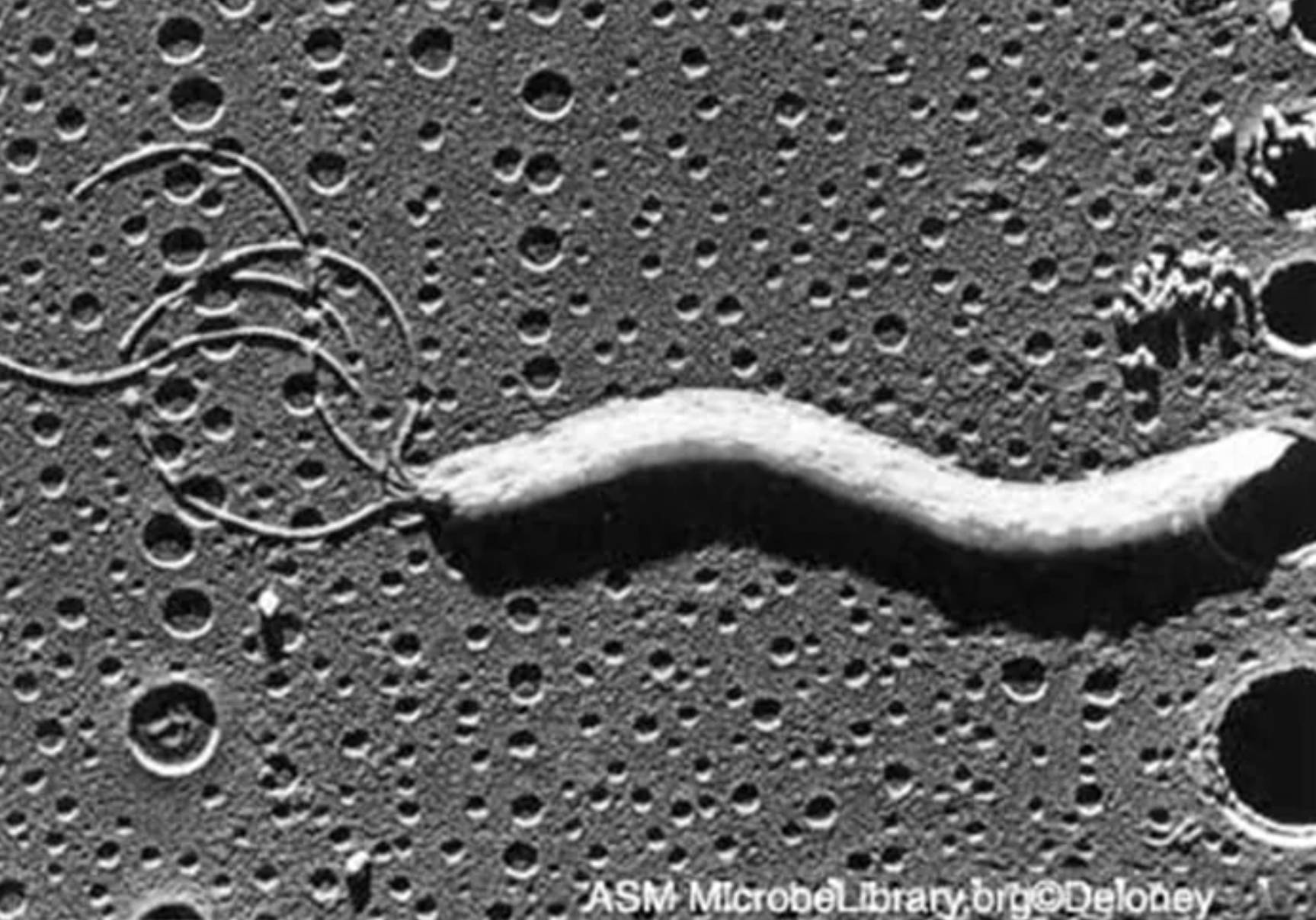
Spiral-shaped bacterium refers to the elongated, S-shaped or helical cell body visible in the center of the image. H. pylori typically measures 0.5 μm in width by 2.5–5 μm in length with one to three gentle spirals, allowing it to corkscrew through viscous gastric mucus. The micrograph shows the characteristic curved morphology and smooth surface of the cell, which is essential for efficient penetration of the protective mucus layer and colonization of the gastric epithelium.
Polar flagella appear as thin, coiled filamentous structures extending from one end of the bacterial cell, often forming a tuft of 4–7 sheathed flagella. These sheathed flagella provide powerful motility in the viscous gastric environment, enabling the bacterium to swim against the mucus flow and reach the epithelial surface. In the image, the flagella are clearly visible as looped or extended threads attached to the cell pole, demonstrating the lophotrichous arrangement critical for its pathogenesis.
Gastric surface forms the pitted, textured background against which the bacterium is positioned. This represents the mucosal or epithelial surface of the stomach lining, characterized by microvilli or mucus layer features that appear as numerous small depressions or pores in the micrograph. The interaction between H. pylori and this surface is key to its ability to adhere and persist in the stomach despite peristalsis and acid secretion.
Morphology and Ultrastructure of Helicobacter pylori
Under electron microscopy, H. pylori displays a classic Gram-negative envelope with an outer membrane, periplasm, and inner membrane. The spiral or S-shaped form is highly motile due to the polar bundle of sheathed flagella, which are covered by an extension of the outer membrane. This sheath protects the flagellar filament in the acidic environment and may play a role in immune evasion. The image beautifully illustrates these features, with the helical cell body and attached flagella standing out against the gastric surface.
The bacterium can switch between spiral and coccoid forms, the latter often seen in stressed or non-culturable states. Spiral forms are associated with active colonization and motility, while coccoid forms may represent a dormant survival strategy. These morphological adaptations allow H. pylori to persist in the stomach for decades if untreated.
- Flagella are sheathed and possess a terminal bulb in some preparations.
- Cell length and spiral turns vary slightly between strains but remain functionally consistent.
- The outer membrane contains lipopolysaccharides with unique Lewis antigens that mimic host structures.
Pathogenesis and Colonization Mechanisms
H. pylori colonizes the gastric mucus layer using its flagella-driven motility to penetrate the viscous barrier and reach the neutral pH zone near the epithelium. Once there, it adheres via adhesins such as BabA and SabA that bind to blood group antigens and sialylated structures on host cells. Urease enzyme hydrolyzes urea to ammonia and carbon dioxide, locally neutralizing gastric acid and creating a protective microenvironment around the bacterium.
Virulence factors including the cag pathogenicity island encode a type IV secretion system that injects the CagA protein into host cells, altering signaling pathways and promoting inflammation. VacA toxin causes vacuolation and apoptosis in epithelial cells. These mechanisms lead to chronic gastritis that can progress to peptic ulcers or malignancy in susceptible individuals.
- Motility is essential for initial colonization and maintenance in the mucus layer.
- Urease is one of the most abundant proteins and a major target for diagnostic tests.
- Chronic infection induces a Th1-dominated immune response that fails to clear the bacterium.
Clinical Significance and Diseases Associated with H. pylori
Infection with H. pylori is a major cause of duodenal and gastric ulcers, accounting for the majority of non-NSAID-related peptic ulcer disease. It is also classified as a class I carcinogen by the WHO due to its strong association with distal gastric adenocarcinoma and MALT lymphoma. Eradication therapy significantly reduces the risk of ulcer recurrence and cancer development in high-risk populations.
Diagnosis relies on non-invasive methods such as urea breath test, stool antigen detection, or serology, and invasive endoscopy with biopsy for histology, culture, or rapid urease test. The spiral morphology observed in the electron micrograph correlates with the organism’s appearance in histological sections stained with Giemsa or Warthin-Starry silver stain.
Treatment typically involves triple or quadruple therapy combining a proton pump inhibitor with antibiotics such as clarithromycin, amoxicillin, or metronidazole. Rising antibiotic resistance, particularly to clarithromycin, necessitates susceptibility testing in refractory cases and the development of new regimens.
- Most infected individuals remain asymptomatic, but a subset develops symptomatic disease.
- Eradication rates have declined due to increasing antimicrobial resistance worldwide.
- Reinfection is uncommon in adults in developed countries but more frequent in high-prevalence areas.
Laboratory Identification and Research Insights
In the laboratory, H. pylori grows slowly on microaerophilic conditions at 37°C on selective media such as chocolate agar or Campylobacter-selective agar. Colonies are small, translucent, and may take 3–7 days to appear. The electron micrograph provides ultrastructural confirmation of identity beyond light microscopy or biochemical tests like urease, catalase, and oxidase positivity.
Research using scanning and transmission electron microscopy has elucidated the bacterium’s interaction with gastric cells, including pedestal formation and cytoskeletal rearrangements induced by CagA. Studies of flagellar structure and motility continue to inform vaccine development and novel therapeutic targets aimed at disrupting colonization.
The image from the ASM MicrobeLibrary highlights the organism’s intimate association with the gastric surface, underscoring why H. pylori is so successful at evading host defenses and persisting long-term. Understanding these morphological and functional features remains essential for advancing diagnostics, treatment, and prevention strategies against this globally significant pathogen.

